When the Brain Goes Offline
From first look to disposition: an ED algorithm for AMS + coma scales + ICP screening
Altered mental status (AMS) is not a diagnosis. It’s a warning signal—sometimes the first and only outward sign of hypoxia, sepsis, intracranial hemorrhage, toxic ingestion, metabolic collapse, or impending respiratory failure. And in the Emergency Department, AMS is also a cognitive trap: it invites broad differentials, scattered testing, delays in definitive therapy, and false reassurance.
What saves patients isn’t doing everything. It’s doing the right things first, in a structured sequence, while continuously reassessing.
This post lays out a practical ED approach to AMS: stabilise physiology, rapidly screen reversible causes, identify structural catastrophe early, and disposition safely. Along the way, we’ll also cover:
GCS vs FOUR score (and when each makes more clinical sense)
ONSD eye POCUS as a bedside screen for raised ICP, with a quick “how I do it” ?
1) AMS in the ED: a syndrome of urgency
AMS accounts for a significant portion of ED presentations and carries high admission rates and meaningful mortality in cohort studies (Kanich, 2002; Xiao, 2012; Cherukuri, 2020). But beyond epidemiology, the operational truth is:
With AMS, you can’t afford a slow workup. You must run physiology and etiology in parallel.
Think of AMS management as two simultaneous tracks:
Track A: stabilise physiology
Oxygenation
Ventilation
Perfusion
Glucose
Temperature
Track B: find the cause
Structural CNS catastrophe?
Toxic/metabolic?
Infection/systemic organ failure?
Seizure/non-convulsive status?
Psychiatric mimic?
This two-track mindset is consistent across ED and prehospital evidence-based guidance (Sanello, 2018; Smith, 2019).
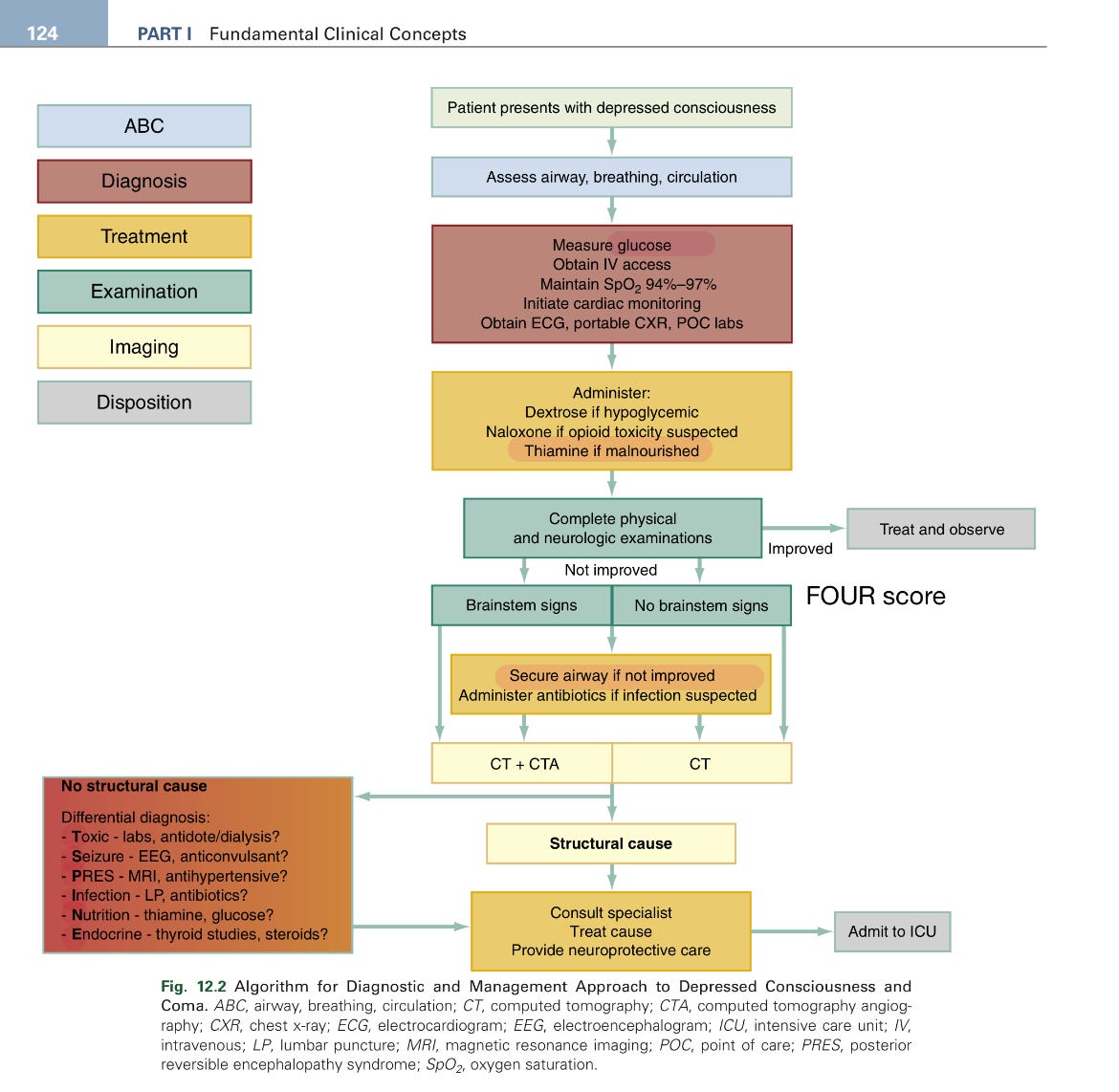
2) First 2 minutes: Stabilise before you intellectualise
Primary survey: ABCD + vitals
Start with airway, breathing, circulation, disability (GCS/alertness) and full vital signs. AMS patients deteriorate quickly and often silently (Han, 2013; Sanello, 2018; Smith, 2019).
Immediate ED actions
Bedside glucose (treat hypoglycaemia immediately)
IV access
Cardiac monitor + ECG
SpO₂ monitoring and oxygen as needed
Temperature (hypo/hyperthermia can mimic neuro catastrophe)
This core bundle is repeatedly reinforced in ED evaluation frameworks (Sanello, 2018; Xiao, 2012).
3) The “coma cocktail” isn’t dead — it’s targeted now
Old teaching often suggests: dextrose + naloxone + thiamine.
Modern practice keeps the spirit but improves the precision:
Dextrose → if hypoglycaemic
Naloxone → if opioid toxicity suspected (respiratory depression, typical toxidrome)
Thiamine → if malnourished, alcohol use disorder, or strong suspicion of deficiency
You’re not giving a ritual. You’re preventing irreversible brain injury while searching for cause (Han, 2013; Smith, 2019).
4) The patient can’t speak — but the story is still there
The AMS history usually comes from the surroundings, not the patient:
EMS and scene narrative (pills, CO exposure, trauma mechanism)
Family/bystanders
Prior medical records
Medication lists
Phone photos of prescriptions
Collateral history is consistently emphasized because it often contains the diagnostic key (Smith, 2019; Sanello, 2018; Ali, 2024).
5) Exam: the highest-yield “test” you already have
One underappreciated finding from ED cohort work is that history and physical examination can outperform broad routine testing for diagnostic yield (Kanich, 2002).
Your exam should aim to:
Identify toxidromes and systemic clues
Find occult trauma
Detect focal neurologic deficits
Look for meningitis/encephalitis clues
Confirm or refute an immediately life-threatening hypothesis
A full neurologic exam should include pupils, gaze, tone, symmetry, reflexes, plantar response, and respiratory pattern.
6) Delirium: the diagnosis we miss most (and pay for later)
In older adults, AMS frequently represents delirium, which is common, serious, reversible—and frequently missed, especially hypoactive delirium (Han, 2013; Wilber, 2016; Odiari, 2015).
A structured mental status exam focusing on attention, orientation, cognition and perception improves recognition (Koita, 2010).
7) The central fork: structural vs non-structural AMS
At the bedside, you must quickly decide whether AMS is likely:
Structural CNS - Focal signs present
Hemorrhage / ischemic stroke with edema
Space-occupying lesions
Hydrocephalus
Trauma
Herniation physiology
Non-structural - B/l signs present
Toxins / medication effects
Metabolic/endocrine crisis
Hypoxia/hypercapnia
Sepsis/systemic failure
Seizure/postictal state
This fork determines imaging urgency, ICU threshold, and neurosurgical involvement.
8) Imaging: CT head is powerful—but only when indicated
CT head should be decisive, not reflexive. Indications commonly include:
Focal deficit
Head trauma
Anticoagulation
New severe headache
Persistent coma / low GCS without explanation
Concern for hemorrhage/herniation
The ACR Appropriateness Criteria supports targeted imaging strategies in AMS/delirium/psychosis contexts (Soares, 2024; Ali, 2024).
9) Testing should be broad—but not blind
Workup should be guided by your structured differential:
Core labs: glucose, electrolytes, renal/hepatic function, CBC, infection markers
ABG/VBG if ventilation/metabolic derangement suspected
Toxicology if relevant
EEG if nonconvulsive status epilepticus suspected
LP if CNS infection is plausible (after appropriate imaging)
Rare causes exist and matter—but are best pursued after stabilization and common killers are excluded (Hagerman, 2022).
GCS vs FOUR Score: Choosing the right “language of coma”
A structured approach to AMS is incomplete without a reliable method to quantify consciousness over time.
The Glasgow Coma Scale (GCS) remains the most used tool in emergency and trauma care because it is simple and universally recognised. But its verbal component becomes unusable in intubated patients, limiting granularity and trend value in many ED/ICU scenarios.
The Full Outline of UnResponsiveness (FOUR) score addresses this gap by removing the verbal element and replacing it with brainstem reflexes and respiratory pattern, making it fully usable in ventilated patients and better able to detect states such as locked-in syndrome, impending herniation, or evolving brain death physiology (Schey, 2024; Pandey, 2024; Ramazani, 2019).
Systematic reviews suggest both scales are valid and reliable; FOUR may have slightly better inter-rater reliability and may offer a modest prognostic edge for mortality in ICU cohorts, though ED studies in medical/non-trauma AMS show broadly comparable performance (Brun, 2024; Schey, 2024; Pandey, 2024). Practically: GCS remains the common language for trauma triage, while FOUR is especially useful when the patient is ventilated and neurological granularity matters.
ONSD Eye POCUS: a bedside screen for raised ICP—useful, not definitive
In ED patients with AMS—especially those with trauma, seizures, stroke symptoms, severe headache, or unexplained depressed consciousness—optic nerve sheath diameter (ONSD) measurement using ocular POCUS provides a rapid, non-invasive screen for elevated ICP while definitive imaging is arranged (Lau, 2023; Blaivas, 2003; Komut, 2016).
It is particularly useful when CT is delayed, unavailable, or when the patient is too unstable to leave the resuscitation bay, helping clinicians prioritise imaging and consider early ICP-mitigating measures (e.g., head elevation, hyperosmolar therapy) in the appropriate clinical context (Lau, 2023; Cannata, 2022).
Meta-analyses and ED data suggest ONSD has high diagnostic accuracy (often around ~90%+ sensitivity/specificity depending on cutoffs), and early ED series showed excellent performance against CT markers of raised ICP (Blaivas, 2003; Lau, 2023). However, ONSD is best viewed as a triage and trend tool, not an endpoint—cutoffs vary, measurements are operator dependent, and it cannot identify the cause of intracranial hypertension, meaning it should never delay CT/MRI, neurosurgical consultation, or invasive monitoring when indicated (Komut, 2016; Lau, 2023). Standardisation efforts, including international consensus quality criteria, reinforce that its value depends on disciplined technique and interpretation, not casual measurement (Hirzallah, 2024).
More About it, read here —> ONSD
Image taken from Rosen’s 10 edition.
Disposition: AMS is often an ICU conversation
Cohort data suggests AMS patients are frequently admitted, with nontrivial mortality (Kanich, 2002; Xiao, 2012; Cherukuri, 2020). A pragmatic ED disposition framework:
ICU: coma, airway risk, unstable vitals, recurrent seizures/status epilepticus, severe derangements, suspected CNS catastrophe
Ward: identified cause needing treatment/monitoring
Observation: transient AMS fully resolved with reassuring evaluation
Discharge (rare): baseline mental status restored, benign reversible cause corrected, reliable support + follow-up
Decision tools exist, but should support—not replace—clinical judgment (Simkins, 2021; Wood, 2022).
AMS isn’t a “workup.”
It’s emergency neuro-resuscitation plus diagnostic discipline—and the quality of your structure directly shapes outcomes.
References
Kanich W, Brady WJ, Huff JS, et al. Altered mental status: evaluation and etiology in the ED. Am J Emerg Med. 2002;20(7):613-617. doi:10.1053/ajem.2002.35464
Xiao H, Wang Y, Xu T, et al. Evaluation and treatment of altered mental status patients in the emergency department. World J Emerg Med. 2012;3(4):270-277. doi:10.5847/wjem.j.1920-8642.2012.04.006
Cherukuri S, Dhanawade V. Altered mental status in the emergency department, a retrospective analysis. Curr Med Issues. 2020;18:300-304. doi:10.4103/cmi.cmi_64_20
Sanello A, Gausche-Hill M, Mulkerin W, et al. Altered Mental Status: Current Evidence-based Recommendations for Prehospital Care. West J Emerg Med. 2018;19:527-541. doi:10.5811/westjem.2018.1.36559
Smith A, Han J. Altered Mental Status in the Emergency Department. Semin Neurol. 2019;39:005-019. doi:10.1055/s-0038-1677035
Han JH, Wilber ST. Altered mental status in older patients in the emergency department. Clin Geriatr Med. 2013;29(1):101-136. doi:10.1016/j.cger.2012.09.005
Wilber ST, Ondrejka JE. Altered Mental Status and Delirium. Emerg Med Clin North Am. 2016;34(3):649-665. doi:10.1016/j.emc.2016.04.012
Odiari EA, Sekhon N, Han JH, David E. Stabilizing and Managing Patients with Altered Mental Status and Delirium. Emerg Med Clin North Am. 2015;33(4):753-764. doi:10.1016/j.emc.2015.07.004
Koita J, Riggio S, Jagoda A. The mental status examination in emergency practice. Emerg Med Clin North Am. 2010;28(3):439-451. doi:10.1016/j.emc.2010.03.008
Hagerman T, Joyce K, Bissonette A. A Review of Rare Etiologies of Altered Mental Status in the Emergency Department. Curr Emerg Hosp Med Rep. 2022:1-12. doi:10.1007/s40138-022-00254-z
Soares BP, Shih RY, Utukuri P, et al. ACR Appropriateness Criteria® Altered Mental Status, Coma, Delirium, and Psychosis: 2024 Update. J Am Coll Radiol. 2024;21(11S):S372-S383. doi:10.1016/j.jacr.2024.08.018
Ali J, Coufal P, Engvall C, Vogel Å. Management of patients with altered mental status in the Emergency Department. Lakartidningen. 2024;121.
Simkins TJ, Bissig D, Moreno G, et al. A clinical decision rule predicting outcomes of emergency department patients with altered mental status. JACEP Open. 2021;2:e12522. doi:10.1002/emp2.12522
Wood L, Delaney M. Is there a right tool for the job? Decision aids and altered mental status in the emergency department. JACEP Open. 2022;3:e12661. doi:10.1002/emp2.12661
Schey J, Schoch M, Kerr D. The Predictive Validity of the Full Outline of UnResponsiveness Score Compared to the Glasgow Coma Scale in the Intensive Care Unit: A Systematic Review. Neurocrit Care. 2024;43:645-658. doi:10.1007/s12028-024-02150-8
Brun F, Fagertun V, Larsen M, Solberg M. Comparison of Glasgow Coma Scale and Full Outline of UnResponsiveness score to assess level of consciousness in ICU and ED: A quantitative systematic review. Aust Crit Care. 2024. doi:10.1016/j.aucc.2024.03.012
Pandey S, Sahu A, Ekka M, et al. Full Outline of UnResponsiveness Score versus Glasgow Coma Scale in Predicting Clinical Outcomes in Altered Mental Status. J Emerg Trauma Shock. 2024;17:102-106. doi:10.4103/jets.jets_76_23
Ramazani J, Hosseini M. Comparison of Full Outline of Unresponsiveness Score and Glasgow Coma Scale in Medical ICU. Ann Card Anaesth. 2019;22:143-148. doi:10.4103/aca.aca_25_18
Lau T, Ahn J, Manji R, Kim D. A Narrative Review of Point of Care Ultrasound Assessment of the Optic Nerve in Emergency Medicine. Life (Basel). 2023;13. doi:10.3390/life13020531
Blaivas M, Theodoro D, Sierzenski P. Elevated intracranial pressure detected by bedside emergency ultrasonography of the optic nerve sheath. Acad Emerg Med. 2003;10(4):376-381. doi:10.1111/j.1553-2712.2003.tb01352.x
Komut E, Kozaci N, Sönmez B, et al. Bedside sonographic measurement of optic nerve sheath diameter as a predictor of intracranial pressure in ED. Am J Emerg Med. 2016;34(6):963-967. doi:10.1016/j.ajem.2016.02.012
Cannata G, Pezzato S, Esposito S, Moscatelli A. Optic Nerve Sheath Diameter Ultrasound to evaluate increased ICP in critically ill pediatric patients. Diagnostics (Basel). 2022;12. doi:10.3390/diagnostics12030767
Hirzallah M, Lochner P, Hafeez M, et al. Optic Nerve Sheath Diameter POCUS Quality Criteria Checklist: International Consensus Statement. Crit Care Med. 2024;52:1543-1556. doi:10.1097/CCM.0000000000006345




This is a really useful way to teach a scary symptom without sensationalizing it! “The brain goes offline” is often the final common pathway of a few physiologic failures, most commonly too little blood flow (perfusion), too little oxygen, or too little metabolic substrate (glucose), and the time course + context usually tells you which bucket you’re in.
What I especially appreciate is the implicit clinical logic: start with ABCs + fingerstick glucose, then ask “Is this syncope (global hypoperfusion), seizure (abnormal network firing), toxic–metabolic (meds, electrolytes, infection), or focal neuro (stroke/ICH)?” That framing keeps people from anchoring on one diagnosis and missing the high-risk ones.
For readers, a few high-yield “don’t miss” signals when someone “checks out”:
1. Exertional syncope, palpitations, chest pain, known structural heart disease, family history of sudden death → think arrhythmia/cardiac outflow until proven otherwise (ECG matters).
2. Persistent confusion, focal weakness/speech changes, severe headache, anticoagulants → treat as neurologic emergency.
3. Tongue bite, prolonged post-ictal period, witnessed rhythmic shaking → seizure workup and safety counseling.
4. And always review the quiet culprits: dehydration/heat, alcohol/cannabis, antihypertensives, diuretics, sedatives, hypoglycemia risk, and sleep deprivation.