VExUS in Acute Care
A Comprehensive Bedside Approach to Venous Congestion
Venous congestion is increasingly recognized as a major determinant of organ dysfunction in critically ill patients. While traditional hemodynamic assessment focuses on arterial parameters, the venous system plays a crucial role in determining organ perfusion.
The Venous Excess Ultrasound (VExUS) score is a point-of-care ultrasound (POCUS) method that integrates inferior vena cava (IVC) size with Doppler assessment of hepatic, portal, and intrarenal veins to quantify systemic venous congestion. By capturing the downstream effects of elevated right atrial pressure (RAP), VExUS provides a dynamic and organ-relevant assessment of hemodynamics .
Physiological Basis
Elevation in RAP is transmitted backward through the venous system, producing characteristic Doppler changes:
Hepatic veins: direct transmission from right atrium
Portal vein: loss of hepatic buffering
Intrarenal veins: end-organ venous congestion
This leads to reduced effective organ perfusion pressure (MAP − CVP) and contributes to organ dysfunction such as acute kidney injury and hepatic congestion .
Read more about doppler USG here - Physics of doppler
Ultrasound Presets and Machine Settings
Accurate VExUS assessment requires appropriate preset selection and Doppler optimization.
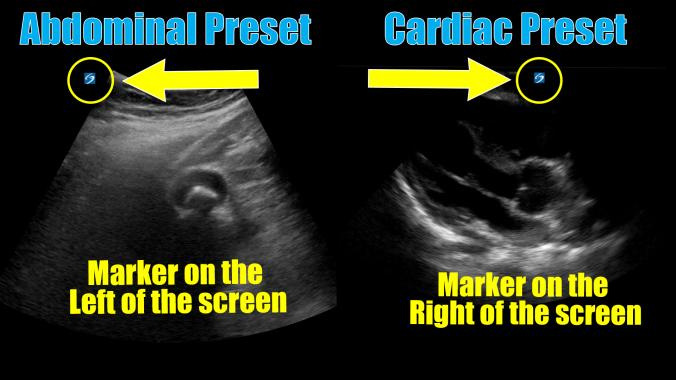
Preset Selection
Cardiac, abdominal, or FAST presets may be used
Cardiac preset allows ECG integration but often requires scale adjustment
Abdominal/FAST presets provide better color Doppler sensitivity
Key Settings
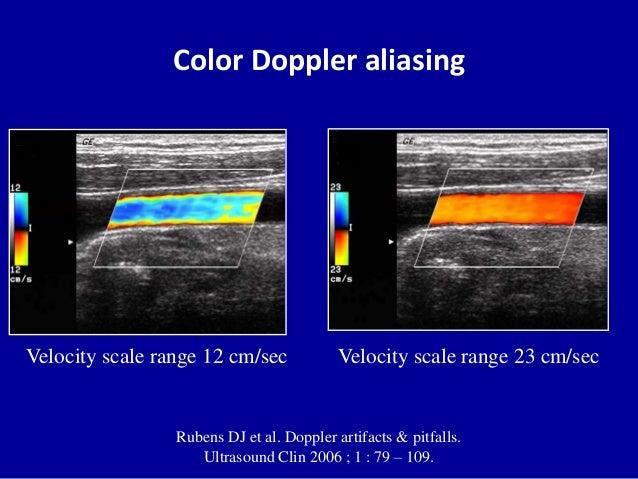
Doppler scale:
~40 cm/s (IVC, HV, PV)
<20 cm/s (intrarenal veins)
Sweep speed: 50–66.7 mm/s
Optimize gain and sample volume
Pitfalls
High scale → no color signal (false low flow)
Low scale → aliasing (false turbulence)
Proper machine setup is essential before interpreting any waveform .
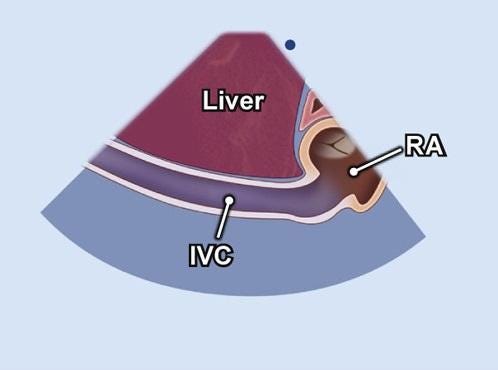
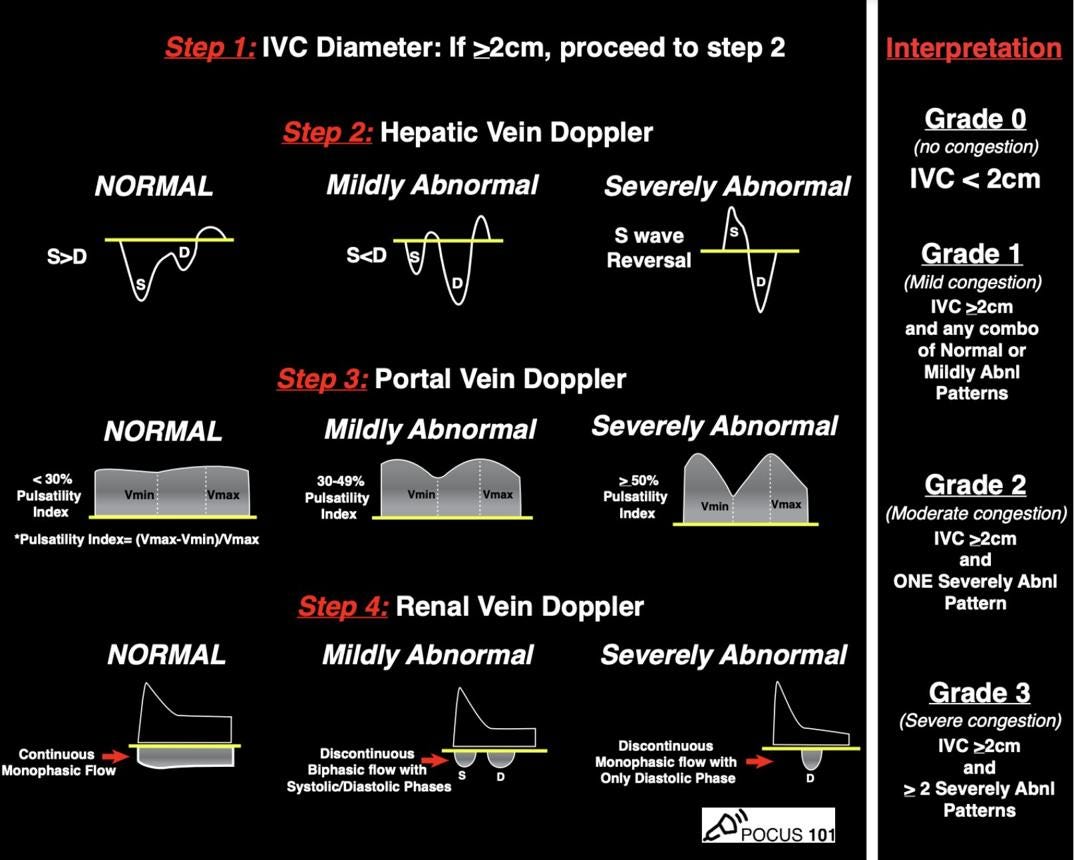
Step 1: Inferior Vena Cava (IVC)
How to Obtain the View
Place probe subxiphoid using liver as window
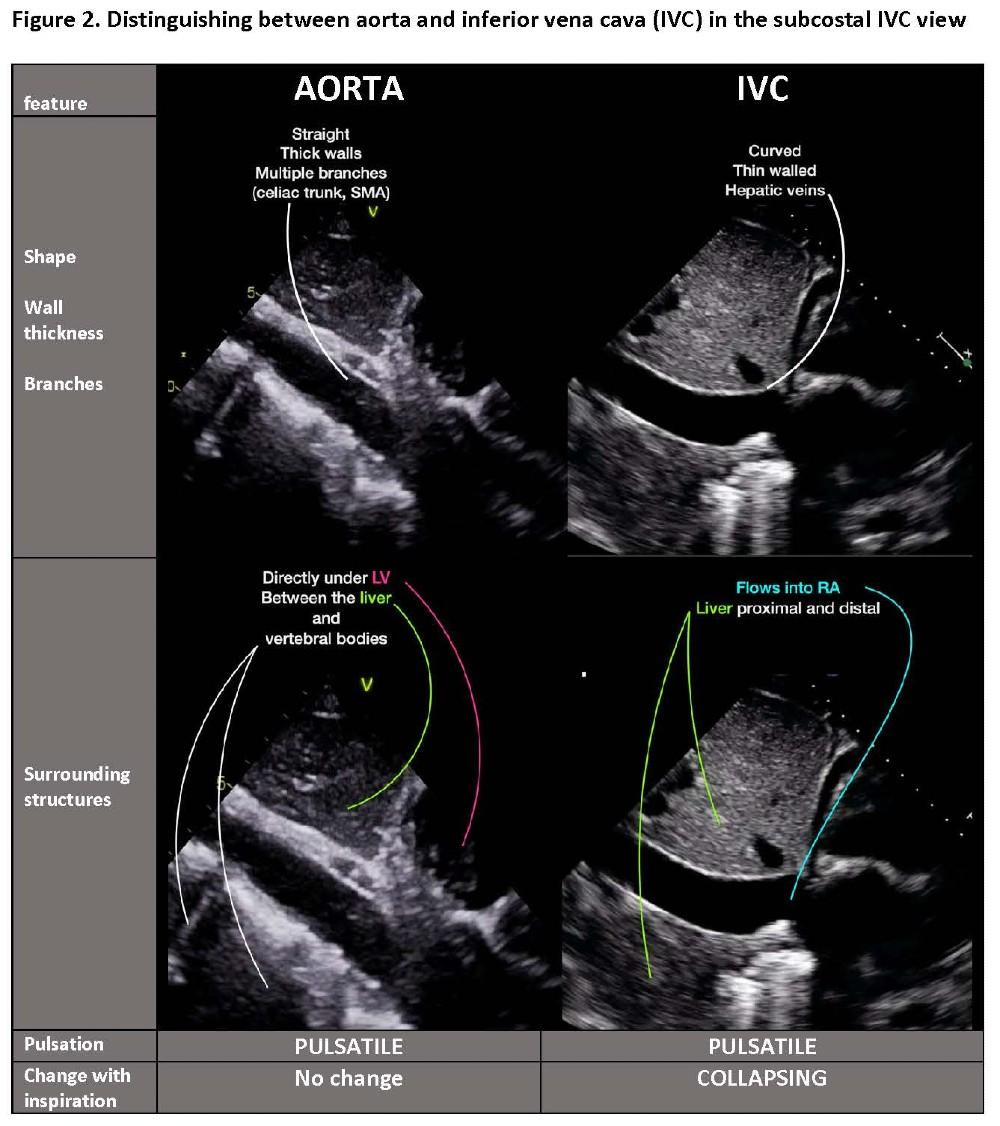
Start with short-axis view (marker at 3 o’clock)
Identify IVC within liver (right side), aorta midline
Rotate to long axis (marker at 12 o’clock)
Visualize IVC entering right atrium and hepatic vein junction
Measurement
2 cm below RA-IVC junction OR
1 cm below hepatic vein junction
Use B-mode, not M-mode
Short-Axis Importance
The short-axis view avoids the “cylinder effect” of long-axis imaging and provides a true cross-sectional representation. It also allows qualitative assessment of a plethoric, circular IVC, which may better reflect elevated RAP, particularly in Asian populations .
Interpretation
≤2 cm → no congestion
≥2 cm → proceed to Doppler
Indian/Asian Considerations
The 2 cm cutoff is widely used but may not be universally applicable. Indian studies apply this threshold without revalidation, while Asian data suggest incorporating collapsibility and morphology for better RAP estimation (Bhardwaj et al., 2020; Khan et al., 2025; Gupta et al., 2023; Assavapokee et al., 2024; Klangthamneam et al., 2024).
Pitfalls
Confusing IVC with aorta
Cylinder effect (long axis underestimation)
Collapsed IVC despite high RAP (raised intra-abdominal pressure)
Dilated IVC in athletes
Misinterpretation using a single view
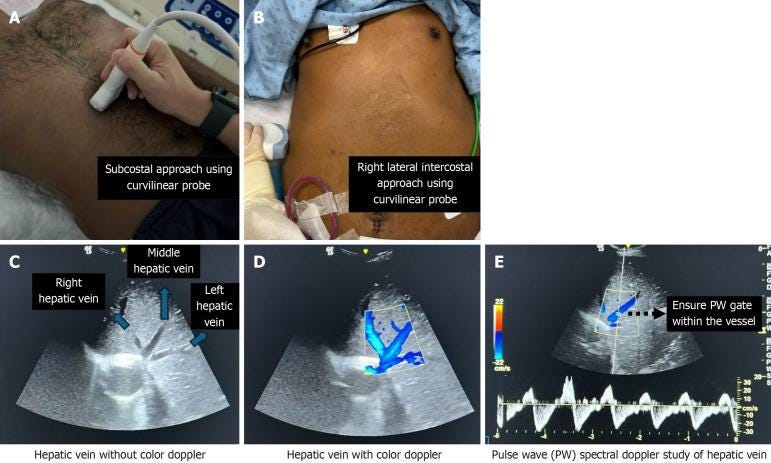
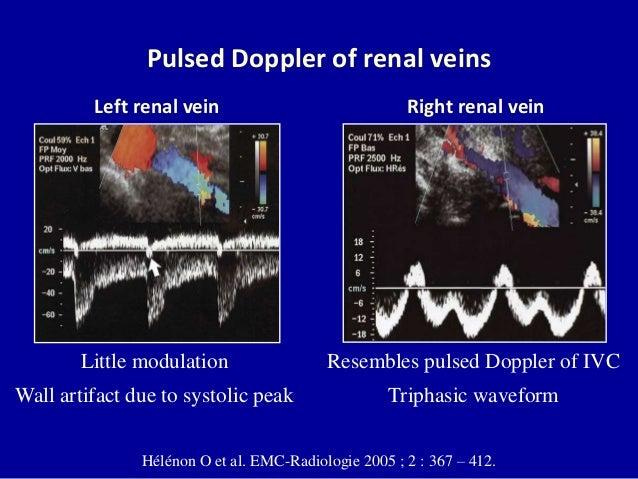
Step 2: Hepatic Vein Doppler
How to Obtain the View
Subxiphoid approach:
Probe below xiphoid, marker at 12 o’clock
Tilt right to visualize hepatic vein entering IVC
Coronal approach (preferred):
Place probe along line from xiphoid to midaxillary line
Marker toward right axilla
Slide cephalad and tilt downward
Doppler technique:
Use color Doppler to identify vein
Place sample volume 1–2 cm from HV-IVC junction
Avoid junctions
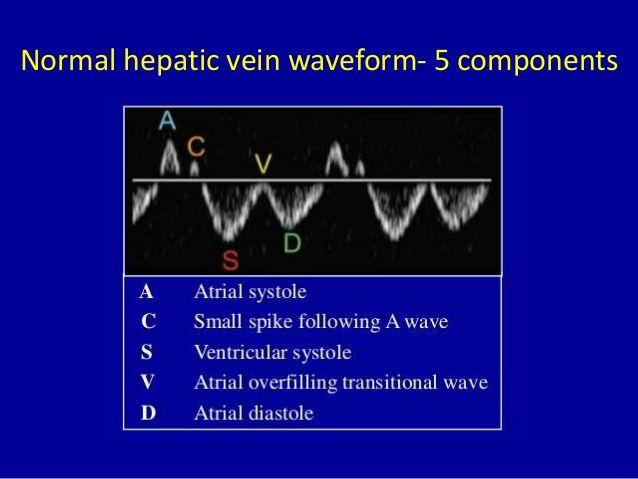
Interpretation
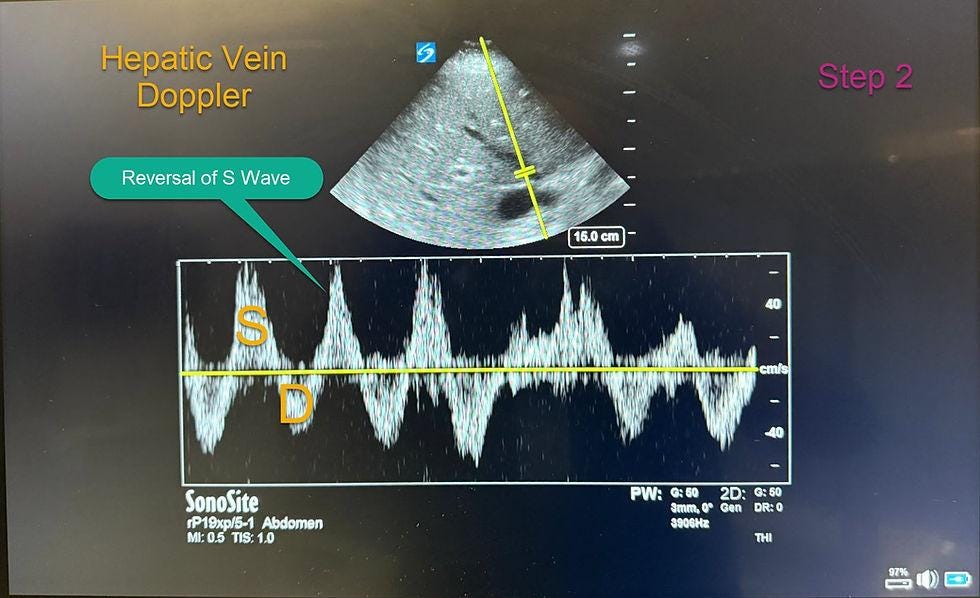
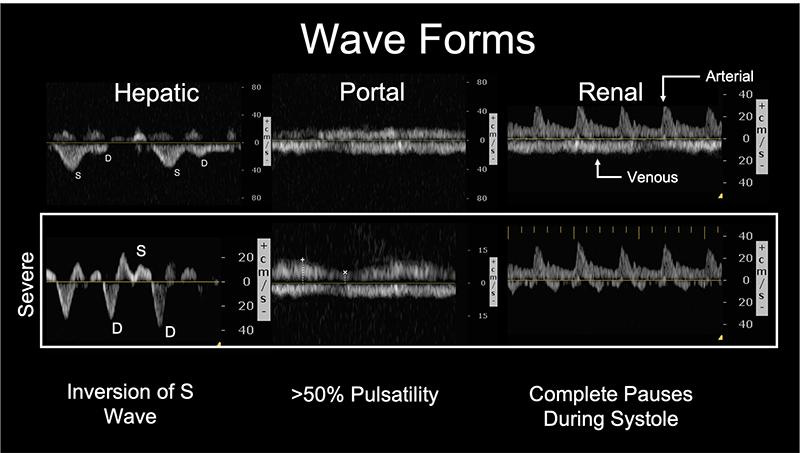
S > D → normal
S < D → mild congestion
S reversal → severe congestion
Pitfalls
Atrial fibrillation (absent A wave)
Tricuspid regurgitation (persistent S reversal)
Cirrhosis (blunted waveform)
Lack of ECG correlation
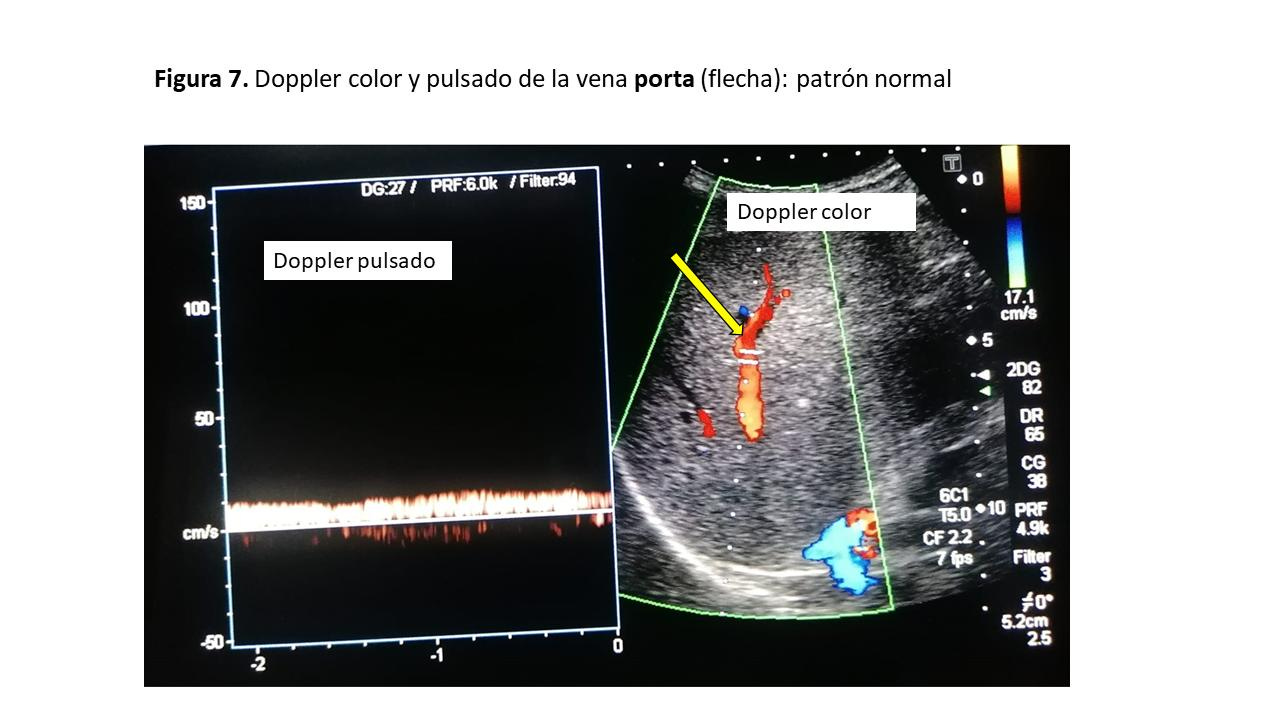
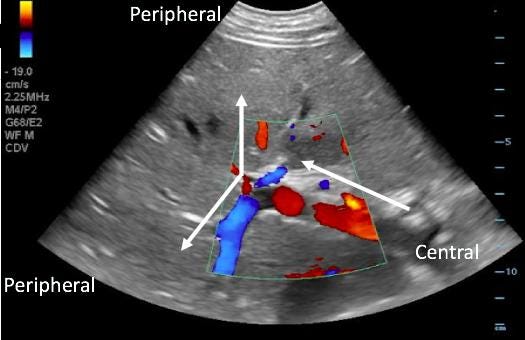
Step 3: Portal Vein Doppler
How to Obtain the View
Use coronal approach similar to hepatic vein
Slide caudally to visualize liver and kidney
Fan anteriorly to identify portal vein
Place Doppler gate in main portal vein (avoid hepatic artery overlap)
Interpretation
PVPF <30% → normal
30–50% → mild
50% → severe
Pitfalls
Cirrhosis altering baseline
Respiratory variation mimicking pulsatility
Hepatic artery contamination
Incorrect gate placement
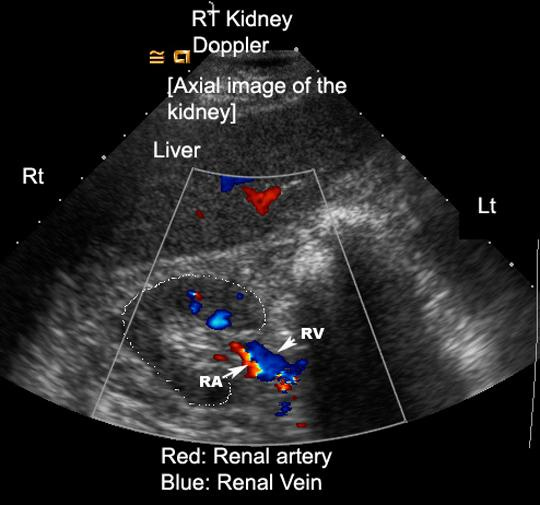
Step 4: Intrarenal Vein Doppler
How to Obtain the View
Coronal approach (xiphoid to posterior axillary line)
Visualize kidney → zoom parenchyma
Use color Doppler to identify interlobar vessels
Place Doppler gate within vein
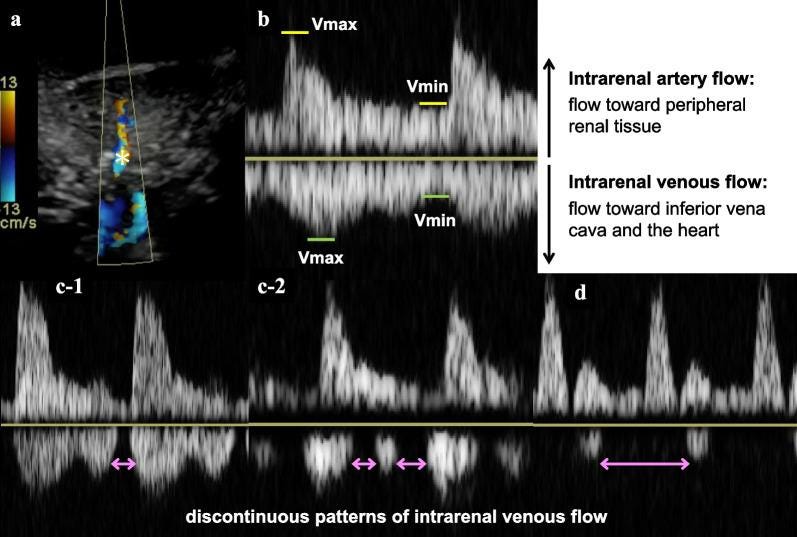
Interpretation
Continuous → normal
Biphasic → mild
Monophasic (D only) → severe
Pitfalls
Technically difficult
Poor visualization in critically ill
Requires low Doppler scale
Limited evidence in CKD
VExUS Grading System
VExUS and Fluid Intolerance
VExUS should be understood as a tool for assessing fluid intolerance, rather than volume status alone. Increasing VExUS grades reflect worsening venous congestion and reduced capacity of the circulation to tolerate additional fluid. Studies have shown that higher VExUS scores correlate with adverse outcomes, including acute kidney injury, and can guide de-resuscitation strategies such as diuretics or ultrafiltration (Jury and Shaw, 2021; Chin et al., 2025; Pierre-Grégoire, 2025; Rola et al., 2024; Bhardwaj et al., 2020). Importantly, VExUS does not predict fluid responsiveness but instead answers whether further fluid administration is likely to be harmful .
Clinical Integration
Not a standalone tool
Must be integrated with cardiac POCUS and clinical context
Does not differentiate volume vs pressure overload
Caution in:
Pulmonary hypertension
Pericardial effusion
Severe tricuspid regurgitation
Conceptual Framework
Venous congestion progresses sequentially:
Right atrium → hepatic veins → portal vein → kidney
The presence of abnormalities further downstream reflects increasing severity of systemic congestion.
Conclusion
VExUS provides a structured, physiologically grounded method for bedside assessment of venous congestion. Its strength lies in integrating multiple venous Doppler signals to reflect organ-level hemodynamics. In Indian and Asian populations, interpretation should emphasize morphology and Doppler findings rather than rigid adherence to IVC diameter thresholds. When applied correctly, VExUS offers a powerful tool to guide fluid management and prevent organ injury.
Read more about views and how to get them here - Decoding VExUS article
Follow @Instagram
References (AMA Style)
Assavapokee T, Rola P, Assavapokee N, Koratala A. Decoding VExUS: a practical guide for excelling in point-of-care ultrasound assessment of venous congestion. Ultrasound J. 2024;16.
Beaubien-Souligny W, Rola P, Haycock K, et al. Quantifying systemic congestion with point-of-care ultrasound: development of the venous excess ultrasound grading system. Ultrasound J. 2020;12(1):16.
Rola P, Haycock K, Spiegel R, et al. VExUS: common misconceptions, clinical use and future directions. Ultrasound J. 2024;16.
Pierre-Grégoire G. VExUS score: optimizing its use in perioperative and critical care management. Crit Care. 2025;29.
Clinical Application & Fluid Intolerance
Jury D, Shaw A. Utility of bedside ultrasound derived hepatic and renal parenchymal flow patterns to guide management of acute kidney injury. Curr Opin Crit Care. 2021;27:587–592.
Chin W, Ngai M, See K. Venous excess ultrasound: a mini-review and practical guide for its application in critically ill patients. World J Crit Care Med. 2025;14.
Bhardwaj V, Vikneswaran G, Rola P, et al. Combination of inferior vena cava diameter, hepatic venous flow, and portal vein pulsatility index (VExUS) in predicting AKI in cardiorenal syndrome. Indian J Crit Care Med. 2020;24:783–789.
De Santamaría C, Fiestas Z, Crespo-Aznarez S, et al. VExUS protocol along cardiorenal syndrome: an updated review. J Clin Med. 2025;14.
Teran S, Mantha A, Siegel G, et al. The role of VExUS in predicting AKI and diuresis in acute decompensated heart failure. Circulation. 2025.
Gravina I, Meo A, La Verde A, et al. VExUS score as a method to guide diuretic treatment in CKD patients with congestive nephropathy. Nephrol Dial Transplant. 2024.
AKI, ICU & Outcomes
Khan W, Saini V, Goel A, Valiyaparambath A. Evaluating the predictive power of VExUS scoring in critically ill noncardiac patients. Indian J Crit Care Med. 2025;29:236–243.
Karki B, Ghimire S, Vaddi B, et al. VExUS: the holy grail or Achilles heel of fluid management? J Nepal Soc Crit Care Med. 2024.
Meshram A, Lalitha A, Philip B, Raj J. Utility of VExUS to quantify venous congestion in critically ill children. Indian J Crit Care Med. 2025;29(Suppl).
Espinosa-Almanza C, Ruiz-Ávila H, Gomez-Tobar J, Acosta-Gutiérrez E. Relationship between cumulative fluid balance and venous congestion using VExUS. J Ultrasound Med. 2025;44.
Utami M, Prawira Y, Djer M, et al. Correlation between fluid balance and VExUS score in critically ill children. BMC Pediatr. 2025;25.
Indian / Regional Context
Gupta B, Ahluwalia P, Gupta A, et al. Utility of VExUS score in perioperative and ICU settings: a systematic review. Indian J Anaesth. 2023;67:S218–S226.
Asian Validation & IVC Considerations
Klangthamneam S, Meemook K, Petnak T, et al. Correlation between right atrial pressure and VExUS, IVC diameter, and jugular venous pressure. Ultrasound J. 2024;16.
Anastasiou V, Peteinidou E, Moysidis D, et al. Multiorgan congestion assessment by VExUS in acute heart failure. J Am Soc Echocardiogr. 2024.
Torres-Arrese M, Mata-Martínez A, Luordo-Tedesco D, et al. Usefulness of systemic venous ultrasound protocols in heart failure prognosis. J Clin Med. 2023;12.
Wong A, Olusanya O, Watchorn J, et al. Utility of VExUS to track dynamic volume status in hemodialysis. Ultrasound J. 2024;16.
Koratala A, Romero-González G, Soliman-Aboumarie H, Kazory A. Unlocking the potential of VExUS in assessing venous congestion. Cardiorenal Med. 2024.
Ahn A, Desai K. Fluid status assessment and the role of VExUS. Tech Vasc Interv Radiol. 2025;28:101026.
Dinesen C, Vistisen S, Aagaard R, et al. Doppler ultrasound assessment of venous congestion for prediction of fluid responsiveness. J Cardiothorac Vasc Anesth. 2024.