The Dose Makes the Poison—even for IV Fluids
Why what you hang, how much you give, and when you stop can decide outcomes in acute care?
Intravenous fluids are among the most frequently prescribed therapies in emergency and acute care—often initiated within minutes of patient contact. Yet, despite their ubiquity, fluids remain one of the most misused interventions in modern medicine. Accumulating evidence now urges us to treat fluids not as benign background therapy, but as pharmacologic agents with indications, dose–response relationships, and toxicity (Myburgh, 2015; Malbrain et al., 2020).
This post explores what fluid to give, when to give it, and—just as importantly—when to stop.
Fluids as Drugs: Indications and Phases of Therapy
Most contemporary frameworks converge on three primary indications for intravenous fluids:
Resuscitation – rapid intravascular volume expansion
Replacement – correction of ongoing or previous losses
Maintenance – provision of daily water and electrolyte needs
A fourth, often under-recognized category is “fluid creep”—fluids delivered unintentionally via drug diluents, line flushes, and nutrition (Malbrain et al., 2020; Stoltzfus et al., 2024).
The R.O.S.E. Concept
The traditional “give fluids early” paradigm has evolved into a dynamic, phase-based model, best represented by the R.O.S.E. framework:
R – Resuscitation: Rapid boluses to restore perfusion
O – Optimization: Fine-tuning stroke volume and cardiac output
S – Stabilization: Avoidance of unnecessary fluids
E – Evacuation: Active de-resuscitation once shock resolves
Failure to transition out of the resuscitation phase leads to fluid accumulation, which has been consistently associated with worse oxygenation, increased acute kidney injury, prolonged ventilation, and mortality (Malbrain et al., 2020; Joannes-Boyau et al., 2022; Mayerhöfer et al., 2022).
Crystalloids vs Colloids: Where the Evidence Landed
For decades, clinicians debated whether colloids were superior due to their theoretical intravascular persistence. Large trials and meta-analyses have largely settled this debate.
Crystalloids: The Default Choice
Crystalloids remain the first-line fluid for most emergency and ICU scenarios, with increasing preference for balanced solutions over 0.9% saline (Myburgh, 2015; Finfer et al., 2018).
Balanced crystalloids reduce:
Hyperchloremic metabolic acidosis
Renal vasoconstriction
Risk of AKI (Mayerhöfer et al., 2022)
Colloids: Volume Efficiency Without Outcome Benefit
While colloids achieve hemodynamic targets with less volume, they do not improve survival compared to crystalloids. More importantly:
Hydroxyethyl starch (HES) is consistently associated with higher rates of AKI and need for renal replacement therapy
Synthetic colloids are now broadly discouraged in critically ill patients (Myburgh, 2015; Lewis et al., 2018; Qureshi et al., 2016)
Albumin occupies a more nuanced position—potentially useful in select populations, but not a universal first-line solution.
Normal Saline vs Balanced Crystalloids: Small Differences, Important Exceptions
The long-running debate between normal saline (NS) and balanced crystalloids (BC) has largely converged on a nuanced conclusion: for most acutely and critically ill adults, average outcome differences are small, but context matters.
Large aggregate and individual-patient meta-analyses show little to no overall mortality difference, with a consistent trend favoring balanced crystalloids (relative risk or odds ratio ~0.93–0.96) and a high probability of modest benefit, particularly in higher-risk patients (Chen & Gao, 2023; Dong et al., 2022; Zampieri et al., 2023).
BC are also associated with slightly lower rates of AKI and major adverse kidney events, an effect most evident in large-volume resuscitation and sepsis cohorts (Self et al., 2018; Hammond et al., 2022; Wang et al., 2023).
However, this population-level advantage does not apply universally: in traumatic brain injury, multiple meta-analyses and IPD data demonstrate higher mortality with balanced crystalloids, making normal saline the preferred fluid in this subgroup (Dong et al., 2022; Zampieri et al., 2023; Diz et al., 2025).
Mechanistically, NS’s supraphysiologic chloride load predisposes to hyperchloremic acidosis and renal vasoconstriction, whereas BC better preserve acid–base balance—but when cerebral osmolality and sodium targets are paramount, saline remains the safer choice (Semler & Kellum, 2019).
In practice, this means balanced crystalloids as default for most ED and ICU patients, with normal saline reserved for clear exceptions, rather than a one-fluid-fits-all approach.
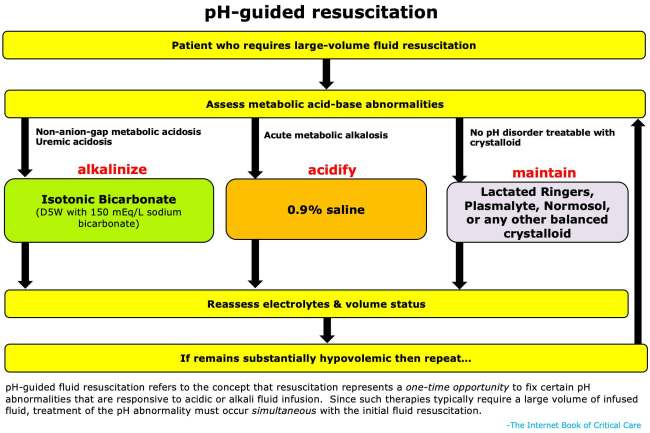
pH-Guided Resuscitation: Using Fluids to Treat Acid–Base Disease
Fluid resuscitation represents a time-limited opportunity to influence pH, particularly early in shock when volume administration is still feasible. Rather than selecting fluids based on sodium concentration or habit, clinicians should consider whether a given fluid will improve or worsen the patient’s acid–base state.
Balanced crystalloids help prevent iatrogenic acidosis; normal saline can correct acute metabolic alkalosis; isotonic bicarbonate can meaningfully treat uremic or non–anion gap metabolic acidosis. Once euvolemia is achieved, further manipulation of pH with fluids becomes unsafe due to volume overload, underscoring the importance of early, intentional fluid choice. In this sense, fluids should be prescribed with the same precision as antibiotics: right drug, right patient, right physiologic target.
more about it @Fluid and pH guided Resuscitation - IBCC
Diagnosis- and Setting-Specific Fluid Choices
General ED and ICU Patients
Balanced crystalloids are favored over saline for most unselected critically ill patients, particularly where large volumes are anticipated (Finfer et al., 2018; Malbrain et al., 2020).
Sepsis and Major Surgery
Network meta-analyses suggest that balanced crystalloids and albumin are associated with lower mortality and AKI compared with saline and HES (Tseng et al., 2020; Lewis et al., 2018).
However, albumin’s cost and lack of clear superiority limit its role to selected patients after initial crystalloid resuscitation.
Trauma (Without TBI)
Modern trauma resuscitation emphasizes damage control:
Isotonic crystalloids in limited volumes
Avoidance of early large-volume resuscitation
Permissive hypotension where appropriate
Early administration of ≥1 L crystalloids has been linked with higher mortality and coagulopathy (McMullan et al., 2024; Goldman et al., 2024; Larson et al., 2025).
Traumatic Brain Injury
TBI is one of the clear exceptions to the “balanced fluid” preference.
Avoid hypotonic fluids and albumin
Normal saline remains preferred, as albumin has been associated with increased mortality in TBI populations (Kruer & Ensor, 2012; Myburgh, 2015; Hammond et al., 2017)
Pediatric Maintenance Fluids
Guidelines now strongly recommend isotonic balanced solutions with added glucose, delivered at restricted rates, to reduce the risk of hyponatremia (Brossier et al., 2022).
Specific Fluid Types
Dextrose Solutions (D5W, D10W, D50W)
Intravenous dextrose solutions serve two distinct purposes: provision of free water and rapid glucose delivery. D5W behaves as free water once glucose is metabolized, making it useful for correcting hypernatremia and providing minimal carbohydrate support, but it is inefficient for hypoglycemia and can precipitate hyponatremia with large volumes. D10W represents the optimal balance for most hypoglycemia management—it delivers effective glucose while remaining safe for continuous peripheral infusion, with less risk of rebound hypoglycemia or venous irritation compared with D50W.
D50W is best reserved for emergent hypoglycemia or scenarios requiring high glucose delivery with minimal volume (e.g., high-dose insulin therapy in beta-blocker or calcium-channel blocker toxicity). However, its extreme hypertonicity increases the risk of extravasation injury and rebound hypoglycemia, favoring D10W when time and access allow. Across all dextrose infusions, clinicians must remain vigilant for hyperglycemia, electrolyte shifts (notably hypokalemia), and thiamine deficiency in malnourished patients.
Lactated Ringer’s (LR)
Lactated Ringer’s is a widely available, inexpensive, and physiologically sound balanced crystalloid. The lactate component is not an acid; it is rapidly metabolized to bicarbonate and may serve as a myocardial fuel. Outside of neurocritical care, LR is appropriate for most ED and ICU patients and is particularly advantageous in acute pancreatitis, DKA, sepsis, and perioperative care.
True contraindications are limited: traumatic brain injury (where evidence favors normal saline), metformin-associated lactic acidosis with impaired lactate clearance, and situations requiring extensive Y-site drug or blood product compatibility. Notably, hyperkalemia and compensated liver disease are not valid contraindications. As with all crystalloids, harm arises from excess volume rather than intrinsic toxicity.
Plasma-Lyte (and Normosol)
Plasma-Lyte is a calcium-free balanced crystalloid buffered with acetate and gluconate, both rapidly converted to bicarbonate. Its main theoretical advantages over LR are broader drug compatibility and utility in patients where calcium administration is undesirable (e.g., hypercalcemia).
Clinically, outcome differences between Plasma-Lyte and LR are negligible, and selection should be guided by local availability, drug compatibility needs, and institutional familiarity, rather than expectation of superior efficacy.
0.9% Sodium Chloride (Normal Saline)
Normal saline remains useful but is best viewed as a specialty fluid rather than a default choice. Its high chloride content promotes non–anion gap metabolic acidosis, renal vasoconstriction, and potassium shifts that can worsen hyperkalemia. These effects are dose-dependent and clinically relevant with moderate to large volumes. Saline retains a role in acute metabolic alkalosis and neurocritical care, particularly traumatic brain injury, where multiple analyses suggest worse outcomes with balanced crystalloids. Outside these indications, routine saline use is difficult to justify on physiologic or evidentiary grounds.
Hypertonic Saline (3% and 23.4%)
Hypertonic saline is a drug, not a maintenance fluid. Its primary indications are symptomatic hyponatremia, cerebral edema, and elevated intracranial pressure. Modern evidence supports the safe peripheral administration of 3% saline, with complication rates lower than those associated with central venous catheter placement. Even 23.4% saline may be given peripherally as a slow bolus in life-threatening neurologic emergencies when rapid ICP reduction is required. Dosing should target a controlled rise in serum sodium (generally 3–5 mM acutely), avoiding overly rapid correction in chronic hyponatremia. Bolus therapy is best supported for emergent ICP control, while continuous infusions risk rebound phenomena once discontinued.
Sodium Bicarbonate (Isotonic and Hypertonic)
Intravenous bicarbonate has specific, physiology-driven indications, not a blanket role in metabolic acidosis. Isotonic bicarbonate (typically prepared in D5W) is particularly useful for uremic metabolic acidosis and non–anion gap metabolic acidosis, where it can improve pH, reduce hyperkalemia, and potentially delay or avoid dialysis. Hypertonic bicarbonate functions as both an alkalinizing and hypertonic agent, making it useful in cerebral edema, severe hyponatremia, and select toxicologic emergencies. Adverse effects are predictable—hypocalcemia, alkalosis, and hypernatremia—and are best mitigated by slow administration and clear physiologic targets.
Albumin
Albumin is not a superior volume expander for most critically ill patients and should not replace crystalloids for routine resuscitation. High-quality evidence supports its use primarily in cirrhosis-related indications: spontaneous bacterial peritonitis, hepatorenal syndrome, and prevention of circulatory dysfunction after large-volume paracentesis. In traumatic brain injury, albumin is associated with increased mortality and should be avoided. Outside liver-specific indications and plasma exchange, albumin adds cost without consistent clinical benefit.
Practical Take-Home Messages for the ED
Fluids are drugs—prescribe with intent
Crystalloids first, balanced solutions when possible
Avoid synthetic colloids, especially HES
Tailor fluid choice to diagnosis (TBI ≠ sepsis ≠ trauma)
Transition early from resuscitation to restriction
Monitor cumulative balance and actively de-resuscitate when appropriate
In acute care, how much fluid you give—and when you stop—matters as much as what fluid you choose.
References
Malbrain MLNG, Langer T, Annane D, et al. Intravenous fluid therapy in the perioperative and critical care setting: executive summary of the International Fluid Academy (IFA). Ann Intensive Care. 2020;10(1):64. doi:10.1186/s13613-020-00679-3
Joannes-Boyau O, Conte L, Bonnet M, et al. Guidelines for the choice of intravenous fluids for vascular filling in critically ill patients. Anaesth Crit Care Pain Med. 2022;41(3):101058. doi:10.1016/j.accpm.2022.101058
Stoltzfus M, Kohli A, Shah P, et al. Narrative review of the role of intravenous fluid in critically sick patients. Egypt J Intern Med. 2024;36:19. doi:10.1186/s43162-024-00301-z
Balanced crystalloids vs normal saline
Chen Y, Gao Y. Balanced crystalloids versus normal saline in critically ill patients: systematic review and meta-analysis. Ther Clin Risk Manag. 2023;19:783-799. doi:10.2147/TCRM.S416785
Dong W, Yan W, Song X, et al. Balanced crystalloids versus normal saline in critically ill patients. Scand J Trauma Resusc Emerg Med. 2022;30:15. doi:10.1186/s13049-022-01015-3
Wang P, Huang Y, Li J, et al. Balanced crystalloid solutions versus normal saline in ICUs: systematic review and meta-analysis. Int Urol Nephrol. 2023;55:2829-2844. doi:10.1007/s11255-023-03570-9
Zampieri FG, Cavalcanti AB, Di Tanna GL, et al. Balanced crystalloids versus saline for critically ill patients (BEST-Living). Lancet Respir Med. 2023. doi:10.1016/S2213-2600(23)00417-4
Self WH, Semler MW, Wanderer JP, et al. Balanced crystalloids versus saline in noncritically ill adults. N Engl J Med. 2018;378(9):819-828. doi:10.1056/NEJMoa1711586
Semler MW, Self WH, Wanderer JP, et al. Balanced crystalloids versus saline in critically ill adults. N Engl J Med. 2018;378(9):829-839. doi:10.1056/NEJMoa1711584
Hammond DA, Lam SW, Rech MA, et al. Balanced crystalloids versus saline in critically ill adults: systematic review and meta-analysis. Ann Pharmacother. 2019;54(1):13-25. doi:10.1177/1060028019866420
Hammond NE, Zampieri FG, Di Tanna GL, et al. Balanced crystalloids versus saline in critically ill adults. NEJM Evid. 2022;1(2). doi:10.1056/EVIDoa2100010
Semler MW, Kellum JA. Balanced crystalloid solutions. Am J Respir Crit Care Med. 2019;199(8):952-960. doi:10.1164/rccm.201809-1677CI
Zwager C, Tuinman PR, de Grooth HJ, et al. Physiology-guided choice between balanced crystalloids and saline. Crit Care. 2019;23:319. doi:10.1186/s13054-019-2658-4
Traumatic brain injury
Diz J, Luna-Rojas P, Díaz-Vidal P, et al. Balanced crystalloids versus saline in critically ill patients with traumatic brain injury. Anesth Analg. 2025. doi:10.1213/ANE.0000000000007368
Hammond NE, Taylor C, Finfer S, et al. Patterns of intravenous fluid resuscitation in ICU patients. PLoS One. 2017;12:e0176292. doi:10.1371/journal.pone.0176292
Lactated Ringer’s (LR)
Maheshwari K, Turan A, Makarova N, et al. Saline versus lactated Ringer’s solution. Anesthesiology. 2020;132(3):614-624. doi:10.1097/ALN.0000000000003130
McIntyre L, Fergusson D, McArdle T, et al. Hospital-wide lactated Ringer’s versus normal saline. N Engl J Med. 2025. doi:10.1056/NEJMoa2416761
De-Madaria E, Herrera-Marante I, González-Camacho V, et al. Lactated Ringer’s versus saline in acute pancreatitis. United European Gastroenterol J. 2018;6(1):63-72. doi:10.1177/2050640617707864
Ocskay K, Mátrai P, Hegyi P, et al. Lactated Ringer’s reduces severity and mortality in acute pancreatitis. Biomedicines. 2023;11:321. doi:10.3390/biomedicines11020321
Zhao T, Kang Z, Zhang Q, et al. Lactated Ringer’s versus saline in acute pancreatitis: meta-analysis. Int J Surg. 2025;111:3467-3480. doi:10.1097/JS9.0000000000002330
Mosquera F, Benítez E, Benavides M, et al. Fluid resuscitation with lactated Ringer vs saline in acute pancreatitis. Diseases. 2025;13:300. doi:10.3390/diseases13090300
Jamison A, Mohamed A, Chedester C, et al. Lactated Ringer’s versus saline in DKA (RINSE-DKA). Pharmacotherapy. 2024;44(6):623-630. doi:10.1002/phar.4600
Agarwal A, Jayashree M, Nallasamy K, et al. Saline versus lactated Ringer’s in pediatric DKA. BMJ Open Diabetes Res Care. 2025;13:e004623. doi:10.1136/bmjdrc-2024-004623
Gelbenegger G, Shapiro NI, Zeitlinger M, et al. Lactated Ringer’s versus saline for sepsis-induced hypotension. Crit Care Med. 2025;53:e1140-e1144. doi:10.1097/CCM.0000000000006601
Kang Y, Ahn S, Kang M. Fluid resuscitation strategies in septic shock: causal inference analysis. Sci Rep. 2025;15:3141. doi:10.1038/s41598-025-03141-1
Ellekjaer K, Perner A, Jensen M, et al. Lactate- versus acetate-buffered crystalloids. Br J Anaesth. 2020. doi:10.1016/j.bja.2020.07.017
Tahir S. Adverse reactions to Ringer’s lactate infusion. Health Sci AUS. 2025. doi:10.70765/ypb55f14
Plasma-Lyte / acetate-buffered solutions
Pfortmueller CA, Faeh L, Müller M, et al. Acetate- versus lactate-buffered balanced solutions in cardiac surgery. Crit Care. 2019;23:159. doi:10.1186/s13054-019-2423-8
Liu X, Cao H, Tan X, et al. Acetate versus lactate Ringer’s in infants with biliary atresia. BMC Pediatr. 2021;21:541. doi:10.1186/s12887-021-03074-4
Hypertonic saline
McKee A, Moriber N, Tornwall J. Intravenous fluid therapy choice in trauma ICU patients. Crit Care Nurse. 2025;45(2):41-49. doi:10.4037/ccn2025318
McMullan J, Curry W, Calhoun D, et al. Fluid resuscitation in trauma: NAEMSP position statement. Prehosp Emerg Care. 2024. doi:10.1080/10903127.2024.2433146
Goldman S, Radomislensky I, Givon A, et al. Early crystalloid resuscitation in trauma. Am J Emerg Med. 2024;88:57-63. doi:10.1016/j.ajem.2024.11.046
Sodium bicarbonate & pH-guided resuscitation
Jaber S, Paugam-Burtz C, Dupont H, et al. Sodium bicarbonate therapy in severe metabolic acidosis (BICAR-ICU). Lancet. 2018;392:31-40. doi:10.1016/S0140-6736(18)31080-8
ARDS Network. Ventilation with lower tidal volumes for ARDS. N Engl J Med. 2000;342:1301-1308. doi:10.1056/NEJM200005043421801
Kraut JA, Madias NE. Treatment of acute metabolic acidosis. N Engl J Med. 2018;379:231-241. doi:10.1056/NEJMra1714704
Albumin
Finfer S, Bellomo R, Boyce N, et al. SAFE study investigators. A comparison of albumin and saline for fluid resuscitation. N Engl J Med. 2004;350:2247-2256. doi:10.1056/NEJMoa040232
Sort P, Navasa M, Arroyo V, et al. Effect of albumin in spontaneous bacterial peritonitis. N Engl J Med. 1999;341:403-409. doi:10.1056/NEJM199908053410603
International Collaboration for Transfusion Medicine Guidelines. Use of intravenous albumin. Transfusion. 2024. doi:10.1111/trf.38447639
Educational / Web resource
Weingart S, Farkas J. Fluid selection & physiology. EMCrit Project. https://emcrit.org/ibcc/fluid/. Accessed 2026.




This is such an important reminder for acute care: IV fluids are a drug with an indication, a dose, a timing window, and predictable adverse effects when we keep “hanging liters” after the physiology has changed. What I loved about your framing is that it pulls people out of the false binary (“fluids good” vs “fluids bad”) and back into systems thinking:
1. Early shock: fluids can be lifesaving when they actually increase effective circulating volume and perfusion.
2. Later shock / post-resuscitation: the same fluid becomes interstitial edema, impaired gas exchange, gut wall edema, renal congestion, worsened wound healing; i.e., harm masquerading as routine.
Clinically, the highest-yield shift is exactly what you’re teaching: fluid stewardship. Give a purposeful bolus, reassess with something dynamic (MAP + pulse pressure variation where appropriate, cap refill, lactate trend, IVC/POCUS, passive leg raise), and then have the courage to stop, and to pivot to vasopressors, source control, or de-resuscitation when the problem is no longer “volume”.
Also appreciate you calling out that “normal saline isn’t neutral.” Chloride load, acid–base effects, and kidney perfusion dynamics are not academic; they show up in real patients.
This is a post that makes teams safer: fewer reflex liters, more deliberate physiology, better outcomes!