I Feel Weak, Doctor.
A practical emergency department framework for evaluating weakness
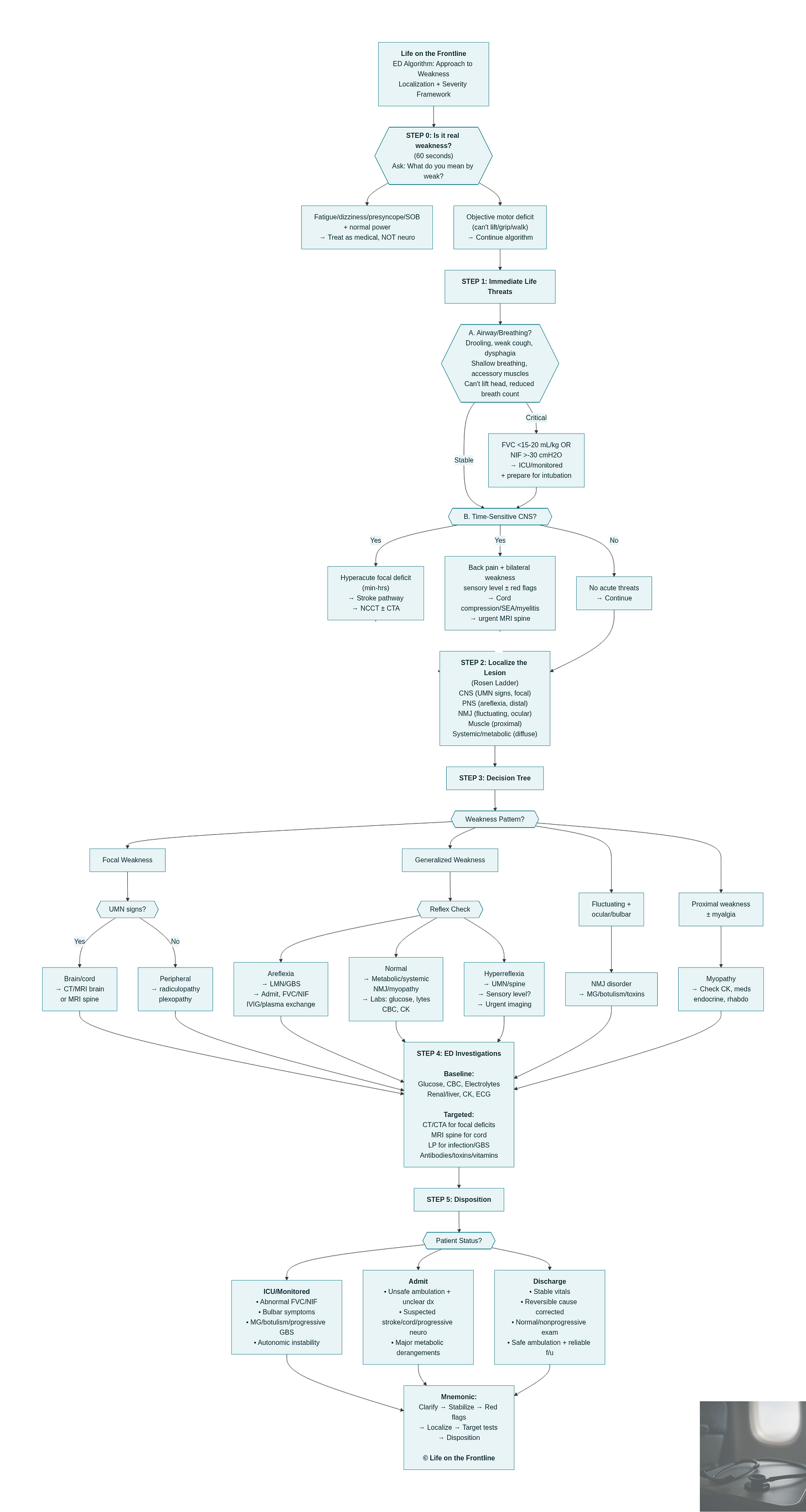
Weakness is one of the most deceptively dangerous chief complaints in Emergency Medicine. It can mean anything from “I feel tired” after a viral illness to catastrophic pathologies like brainstem stroke, spinal cord compression, Guillain–Barré syndrome (GBS), and myasthenic crisis.
Our job in the ED isn’t to label every case immediately — it’s to stabilize, localize, and recognize time-critical causes early enough to change outcomes (Khamees & Meurer, 2021; Sams et al., 2025; Ganti & Rastogi, 2016; Walls et al., 2023).
Step 1: Clarify what the patient means by “weakness”
Many patients use “weak” as shorthand for:
fatigue
dizziness
presyncope
shortness of breath
general malaise
ataxia
But neurologically, weakness means:
Reduced ability to generate normal muscle force, up to paralysis.
The first question is always:
“What exactly do you mean by weak?”
Ask them to describe tasks rather than symptoms:
“Can you get out of bed?”
“Can you climb stairs?”
“Are you dropping objects?”
“Any difficulty walking?”
This single step separates true motor deficit from nonspecific complaints and prevents premature neuro workups (Khamees & Meurer, 2021; Nickel et al., 2009; Walls et al., 2023).
Practical split
True motor weakness vs fatigue/presyncope
Focal vs generalized
Acute vs subacute vs chronic
(Khamees & Meurer, 2021; Vasconcelos et al., 2017)
Step 2: Immediate ED priorities (before localization)
Before we get elegant with neuroanatomy, two questions dominate:
1) Is this patient about to stop breathing?
Neuromuscular weakness can quietly progress into respiratory failure.
Look for:
shallow rapid respirations
inability to manage secretions
bulbar dysfunction (dysphagia/dysarthria)
inability to lift head off bed
weak cough / pooling saliva
In suspected GBS / myasthenia / botulism, measure:
Vital capacity (VC / FVC)
Maximal inspiratory pressure (MIP / NIF)
High-risk thresholds used in EM practice:
FVC < 15–20 mL/kg
MIP (NIF) between 0 and –30 cmH₂O
These should trigger elective intubation consideration or at least monitored/ICU disposition (Khamees & Meurer, 2021; Sams et al., 2025; Morgan, 2015; Walls et al., 2023).
2) Is there time-sensitive CNS disease?
Weakness with a neuro exam abnormality is stroke until proven otherwise, and weakness with definite sensory level/back pain is cord compression until proven otherwise.
Hyperacute focal weakness
Think:
ischemic stroke / hemorrhage
brainstem infarct
large vessel occlusion
(Khamees & Meurer, 2021)
Weakness + back pain + red flags
Think:
epidural abscess
epidural hematoma
malignant cord compression
transverse myelitis
Red flags:
cancer
fever
IV drug use
recent spine procedure
trauma
(Vasconcelos et al., 2017; Walls et al., 2023)
Important airway nuance
In prolonged neuromuscular illnesses (>3 days), avoid succinylcholine due to hyperkalemia risk. Prefer non-depolarising paralytics such as rocuronium/vecuronium (Khamees & Meurer, 2021; Walls et al., 2023).
Step 3: The Rosen’s-style localization ladder
Once the patient is stable, the most reliable ED framework is anatomical localization, moving from:
Brain → Spinal cord → Roots/plexus → Peripheral nerve → NMJ → Muscle → Systemic/metabolic
This is the classic emergency neurology approach: it narrows differentials early, prevents shotgun testing, and allows the ED clinician to prioritize dangerous diagnoses before rare ones (Khamees & Meurer, 2021; Sams et al., 2025; Ganti & Rastogi, 2016; Walls et al., 2023)
Unilateral vs bilateral weakness: the first localization fork
One of the fastest ways to narrow the differential in the ED is to classify weakness as unilateral or bilateral. Unilateral weakness (hemiparesis)—especially when abrupt in onset—should be treated as stroke until proven otherwise, particularly if it follows a vascular pattern and is accompanied by cortical signs such as aphasia, neglect, visual field deficits, or gaze deviation; this warrants urgent neuroimaging and reperfusion consideration based on institutional stroke pathways (Khamees & Meurer, 2021; Sams et al., 2025; Walls et al., 2023).
In contrast, bilateral weakness more commonly reflects spinal cord disease, peripheral neuropathy, neuromuscular junction pathology, muscle disease, or systemic/metabolic illness.
—>Bilateral weakness with UMN signs (hyperreflexia, spasticity, Babinski) or a sensory level strongly suggests a spinal cord process such as compression, transverse myelitis, or epidural abscess, and should prompt urgent MRI spine—especially when red flags (back pain, fever, malignancy, IV drug use, recent spinal procedures) are present (Khamees & Meurer, 2021; Vasconcelos et al., 2017; Walls et al., 2023).
—>Bilateral weakness with LMN features (areflexia, low tone) shifts concern toward acute polyneuropathy such as GBS, where respiratory monitoring is as important as limb examination (Sams et al., 2025; Morgan, 2015). Finally, bilateral “weakness” with normal objective power is often systemic/metabolic rather than neurologic, reinforcing the value of clarifying the complaint before escalating to extensive neuroimaging (Ganti & Rastogi, 2016; Nickel et al., 2009).
Ascending vs descending quadriparesis: a pattern that narrows the differential fast
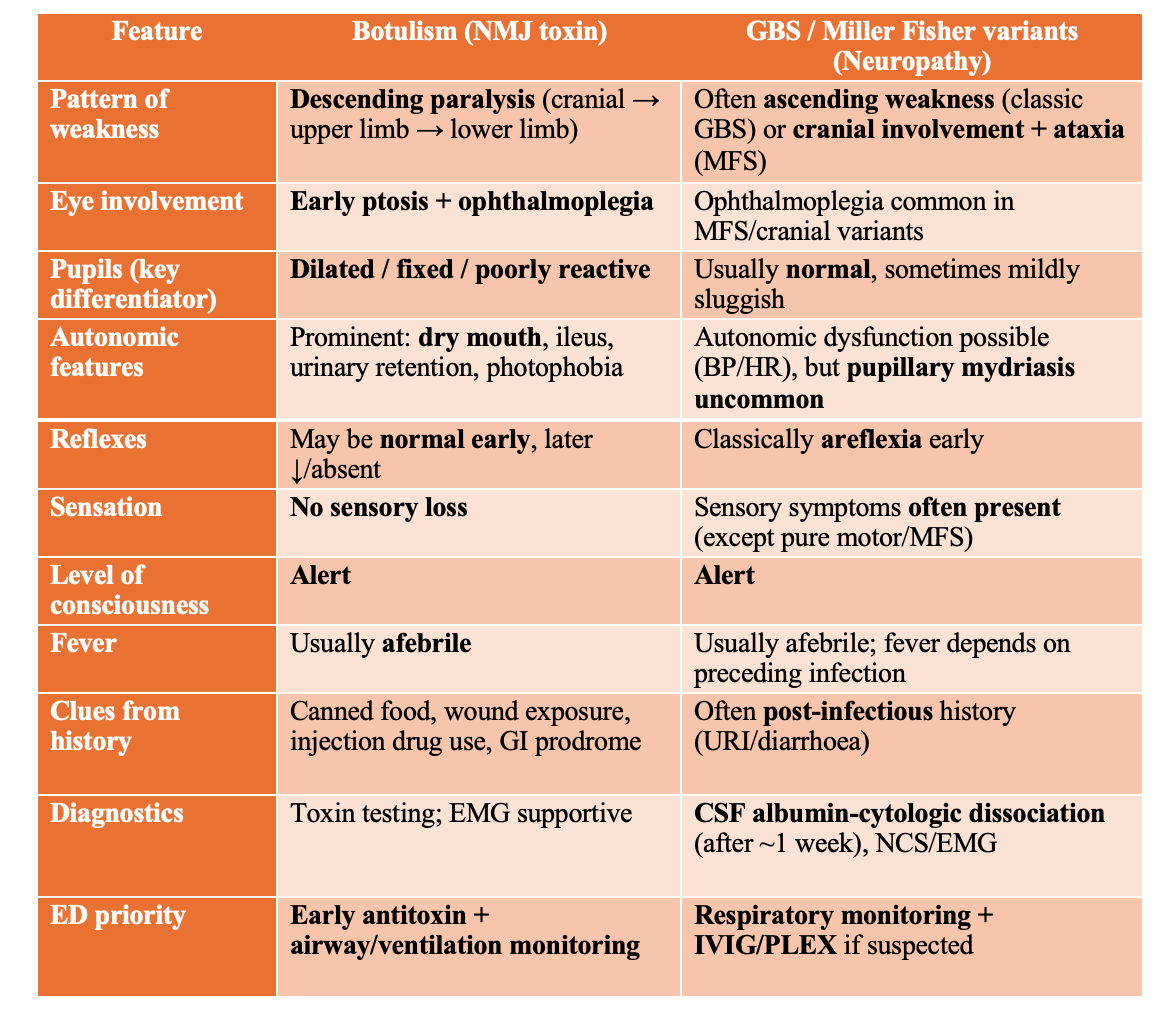
When weakness involves all four limbs (quadriparesis), the direction of progression often provides the diagnosis before the labs do. Ascending quadriparesis—starting in the legs and moving upward—classically suggests acute inflammatory polyradiculoneuropathy (GBS). Patients develop progressive symmetric weakness with areflexia, often after a recent infection; the critical ED priority is serial respiratory assessment because diaphragmatic weakness may evolve quickly even when limb weakness appears mild (Khamees & Meurer, 2021; Sams et al., 2025; Morgan, 2015; Walls et al., 2023).
In contrast, descending quadriparesis—beginning with cranial nerve or bulbar symptoms (ptosis, diplopia, dysarthria, dysphagia) and then progressing to neck, upper limbs, and finally the legs—should immediately raise suspicion for botulism and other toxin-mediated neuromuscular disorders. This can mimic brainstem stroke early; pupillary abnormalities, dry mouth, autonomic symptoms, or exposure history (canned foods, wound contamination, injection drug use) strengthen the diagnosis (Ganti & Rastogi, 2016; Khamees & Meurer, 2021).
A third high-yield pattern is fatigable generalized weakness with ocular/bulbar involvement and fluctuating severity, pointing strongly to myasthenia gravis, where crisis is defined by respiratory/bulbar failure rather than limb power alone (Khamees & Meurer, 2021; Sams et al., 2025).
In all three scenarios, ED management should anchor around airway vigilance and objective respiratory measurements (FVC/NIF), because exam appearance can underestimate impending ventilatory failure (Khamees & Meurer, 2021; Walls et al., 2023).
Step 4: The bedside localization map
A) CNS (brain / brainstem / spinal cord)
Classic clues:
UMN signs: hyperreflexia, spasticity, Babinski
patterned deficit: hemiparesis, sensory level
sphincter involvement for cord lesions
Brain/brainstem: cranial nerve signs, ataxia, dysarthria, altered mental status (Khamees & Meurer, 2021).
Spinal cord: paraparesis/quadriparesis + sensory level ± urinary retention (Khamees & Meurer, 2021; Walls et al., 2023).
B) Peripheral nerve / roots
Classic clues:
LMN signs (low tone, areflexia)
distal > proximal weakness
sensory loss common
GBS remains the archetype:
progressive weakness
often ascending
areflexia
autonomic instability possible
can present in variants too with facial nerve involvement.
Early recognition matters: respiratory decline can be sudden, and time to immunotherapy is clinically important (Khamees & Meurer, 2021; Morgan, 2015; Sams et al., 2025).
C) Neuromuscular junction
Signature clues:
fatigability
fluctuating weakness
ocular + bulbar involvement
Myasthenia gravis
ptosis/diplopia
dysarthria/dysphagia
proximal limb weakness
worsens with activity, improves with rest
A myasthenic crisis is defined less by limb weakness and more by bulbar/respiratory compromise, so serial FVC/NIF monitoring is critical (Khamees & Meurer, 2021; Sams et al., 2025; Walls et al., 2023).
Botulism
descending flaccid paralysis
possible pupillary involvement
can resemble basilar stroke early
(Ganti & Rastogi, 2016)
D) Muscle
Typical clues:
symmetric proximal weakness
myalgia possible
CK often elevated
Think:
rhabdomyolysis
inflammatory myopathy
endocrine myopathy
drug-induced myopathy (statins, steroids)
(Ganti & Rastogi, 2016; Khamees & Meurer, 2021)
E) Systemic / metabolic
The most common “ED weakness” bucket:
sepsis
electrolyte derangements (K/Ca/Na)
endocrine disorders (thyroid, adrenal)
anemia
nutritional deficiencies (B1/B6/B12, vitamin E)
(Ganti & Rastogi, 2016)
In older adults, this complaint is associated with meaningful morbidity and can hide serious disease (Herzog et al., 2020; Nickel et al., 2009).
Step 5: Rational ED investigations (targeted, not shotgun)
Baseline tests (most unwell or unclear cases)
bedside glucose
CBC
electrolytes (Na/K/Ca)
renal + liver function
CK
ECG
± cultures/ABG/VBG if sepsis/resp compromise
(Ganti & Rastogi, 2016; Khamees & Meurer, 2021)
Imaging
CT head (± CTA) if stroke/bleed/LVO suspected (Khamees & Meurer, 2021)
MRI spine if cord compression/transverse myelitis/epidural abscess suspected (Vasconcelos et al., 2017; Walls et al., 2023)
Advanced tests
LP (GBS, meningitis, myelitis) (Khamees & Meurer, 2021)
EMG/NCS (usually inpatient)
AChR/MuSK antibodies, toxin screens, vitamin levels guided by suspicion
(Sams et al., 2025)
Step 6: Management and disposition
Weakness patients are often under-triaged because the complaint sounds nonspecific. But outcomes data shows weakness/fatigue presentations may carry significant risk, especially in older adults (Herzog et al., 2020; Nickel et al., 2009).
ICU / monitored care
abnormal FVC/NIF
bulbar symptoms
suspected MG crisis / botulism / progressive GBS
autonomic instability
Admit
uncertain diagnosis + unsafe ambulation
suspected cord pathology / stroke / evolving neuro deficits
significant metabolic derangements needing correction
Discharge (only if all true)
stable vitals
clear reversible cause identified and corrected
stable, non-progressive symptoms
safe ambulation + reliable follow-up + strict return precautions
Special Mention: Potassium and hypo-glycemia as the “reversible paralysis” diagnosis
For the emergency clinician, potassium deserves a special place in the weakness differential because it represents one of the most rapidly reversible causes of acute flaccid paralysis—yet also one of the most lethal if missed due to malignant arrhythmias. The key bedside clue is that potassium-related weakness is often painless, predominantly motor, and typically spares sensation, and may present dramatically as quadriparesis with preserved consciousness, mimicking GBS, spinal cord disease, or even stroke (Kim et al., 2023; Howard et al., 2025; Jain et al., 2025). Therefore, serum potassium and an ECG should be considered “must-not-miss tests” in any patient presenting with acute generalized weakness or acute flaccid paralysis.
In parallel, bedside glucose must be checked immediately, because hypoglycemia is another rapidly reversible cause of apparent neurologic deficit—capable of presenting with confusion, focal deficits, seizure, or stroke-mimic weakness—and delayed recognition can cause irreversible harm. When identified early, correction of the potassium abnormality and/or hypoglycemia (along with identification of triggers such as thyrotoxicosis, renal tubular acidosis, GI/renal losses, CKD, or potassium-altering drugs) typically leads to near-complete neurologic recovery and prevents recurrence (Kim et al., 2023; Elliott & Braun, 2017; Kreitzer et al., 2025; PoudelJaishi et al., 2022).
Botulism vs GBS : Quick Differentiation
References
Sams W, Hassan N, Meurer W. General approach to weakness. Emerg Med Clin North Am. 2025;44(1):219-226. doi:10.1016/j.emc.2025.08.013
Khamees D, Meurer W. Approach to acute weakness. Emerg Med Clin North Am. 2021;39(1):173-180. doi:10.1016/j.emc.2020.09.010
Vasconcelos M, Vasconcelos L, Brito A. Assessment of acute motor deficit in the pediatric emergency room. J Pediatr (Rio J). 2017;93(Suppl 1):26-35. doi:10.1016/j.jped.2017.06.003
Sanden J, Dryver E. Approach to weakness in the emergency department. Lakartidningen. 2020;117.
Ganti L, Rastogi V. Acute generalized weakness. Emerg Med Clin North Am. 2016;34(4):795-809. doi:10.1016/j.emc.2016.06.006
Latronico N, Rasulo F, Eikermann M, Piva S. Critical illness weakness, polyneuropathy and myopathy: diagnosis, treatment, and long-term outcomes. Crit Care. 2023;27:—. doi:10.1186/s13054-023-04676-3
Herzog S, Jenny M, Nickel C, Ortega N, Bingisser R. Emergency department patients with weakness or fatigue: can physicians predict their outcomes at the front door? A prospective observational study. PLoS One. 2020;15(10):e0239902. doi:10.1371/journal.pone.0239902
Nickel C, Nemec M, Bingisser R. Weakness as presenting symptom in the emergency department. Swiss Med Wkly. 2009;139(17-18):271-272.
Morgan L. The child with acute weakness. Clin Pediatr Emerg Med. 2015;16(1):19-28. doi:10.1016/j.cpem.2015.02.004
Calhoun E, Shih R, Hughes P, Solano J, Clayton L, Alter S. Head computerized tomography in emergency department evaluation of the geriatric patient with generalized weakness. J Am Coll Emerg Physicians Open. 2023;4:—. doi:10.1002/emp2.12998
Vanhorebeek I, Latronico N, Van den Berghe G. ICU-acquired weakness. Intensive Care Med. 2020;46:637-653. doi:10.1007/s00134-020-05944-4
Shimoni Z, Firas A, Hermush V, Froom P. Older hospitalised patients with a chief complaint of weakness and nonspecific presentations are not at risk of adverse health outcomes. J Eval Clin Pract. 2024;31:—. doi:10.1111/jep.14183
McQuown C, Tsivitse E. Nonspecific complaints in older emergency department patients. Clin Geriatr Med. 2023;39(4):491-501. doi:10.1016/j.cger.2023.04.007
Hermans G, Van den Berghe G. Clinical review: intensive care unit acquired weakness. Crit Care. 2015;19:—. doi:10.1186/s13054-015-0993-7
Cavaliere G, Murali N, Bontempo L, Dezman Z. 19-year-old woman with intermittent weakness. Clin Pract Cases Emerg Med. 2021;5:276-282. doi:10.5811/cpcem.2021.4.52011
Anderson R, Hallen S. Generalized weakness in the geriatric emergency department patient: an approach to initial management. Clin Geriatr Med. 2013;29(1):91-100. doi:10.1016/j.cger.2012.10.002
Walls RM, Hockberger RS, Gausche-Hill M, eds. Rosen’s Emergency Medicine: Concepts and Clinical Practice. 10th ed. Philadelphia, PA: Elsevier; 2023.
Dankar R, Barakat S, El-Charabaty E, Hashmi S, Sayegh S. Unraveling an uncommon encounter: hypokalemic periodic paralysis with Brugada phenocopy amidst hypokalemia. Eur J Case Rep Intern Med. 2025;12:—. doi:10.12890/2025_005195
Mulyono R, Hasyim H. A case report of 29-years-old man with hypokalemic periodic paralysis. Magna Neurologica. 2025;3(2):—. doi:10.20961/magnaneurologica.v3i2.1683
Puspamaniar V, Firdha A, Azizah N, Hidayati H. Neurological manifestations of hypokalemia: a case report. Anaesth Pain Intensive Care. 2024;28(1):—. doi:10.35975/apic.v28i1.2387
Anilbhai P, Anuradha N, Prabhu V, Jayaraman V, Vignesh K. Hyperkalemia causing severe muscle weakness in a patient with renal insufficiency. Gomal J Med Sci. 2021;19(3):—. doi:10.46903/gjms/19.03.1034
Wani M, Marchant Z, Nadir U. Hypokalemic periodic paralysis: a case report. Cureus. 2025;17:—. doi:10.7759/cureus.94524
Kim M, Valerio C, Knobloch G. Potassium disorders: hypokalemia and hyperkalemia. Am Fam Physician. 2023;107(1):59-70.
[Unattributed author] Laporan kasus hipokalemi periodik paralisis pada pasien dengan Graves’ disease. Jurnal Penyakit Dalam Indonesia. 2025;12(2):—. doi:10.7454/jpdi.v12i2.1500
Dao K, Dhillon A, Uddin S, Eppanapally S, Lai H. A unique case of a mildly symptomatic patient with severe hypokalemia secondary to furosemide. J Investig Med High Impact Case Rep. 2024;12:—. doi:10.1177/23247096241300932
Jain M, Mehta G, Parmar A, Shastri M, Patel S, Gandhi V, Karatela S, Patel A. Hypokalemic paralysis is not always periodic: a case series. Case Rep Med. 2025;2025:—. doi:10.1155/carm/9925534
Kreitzer N, Albert N, Amin A, et al. EMCREG—International Multidisciplinary Consensus Panel on management of hyperkalemia in chronic kidney disease and heart failure. Cardiorenal Med. 2025;15:133-152. doi:10.1159/000543385
Howard R, Baheerathan A, Brown R, Spillane J, Waraich M. Neurological aspects of electrolyte disorders. Pract Neurol. 2025;25:303-312. doi:10.1136/pn-2023-003801
Delimaris D. Hypo- and hypernatremia, hypo- and hyperkalemia, hypo- and hypercalcemia in human plasma: causes, manifestations and treatment. ARC J Surg. 2024;10(2):—. doi:10.20431/2455-572x.1002001
Sarahfebi I, Nadya A, Lisna A, Anjali P, Rahardjo T, Moenardi V. Paralysis due to hypokalemia and hypocalcemia: a case report. Medical Clinical Update. 2022;1(1):—. doi:10.58376/mcu.v1i1.11
Joshi D, Samadhiya S, Mishra N. All flaccid paralysis is not GBS, some are ??? A case report and systematic review. Int J Nutr Pharmacol Neurol Dis. 2025. doi:10.4103/ijnpnd.ijnpnd_6_25
PoudelJaishi P, Neupane S, Neupane P. Case report: hyperthyroid hypokalemic periodic paralysis. Ann Med Surg (Lond). 2022;78:103759. doi:10.1016/j.amsu.2022.103759
Ambalkar G, Arya N, Ratnani G, Raghuveer R, Saklecha A. Physiotherapy strategies in hypokalemic periodic paralysis: a case report. Cureus. 2024;16:—. doi:10.7759/cureus.52294
Elliott T, Braun M. Electrolytes: potassium disorders. FP Essent. 2017;459:21-28.
Varma V, Patel A, Pathak N, et al. Hypokalemic periodic paralysis in a patient with primary Sjögren’s syndrome and distal renal tubular acidosis: a case report. Clin Med Insights Case Rep. 2025;18:—. doi:10.1177/11795476251372407
Angirekula S, Bhellum P, Rohila A, Dholavan M. An unusual case of drug-induced hyperkalemia presenting as ascending paralysis: a case report and review of literature. Egypt J Intern Med. 2025;37:—. doi:10.1186/s43162-025-00515-9