FFP Transfusion in acute care: When to Give, When to Hold, and Why It Matters ?
Beyond the INR: Evidence-Based Use in Acute Care
Fresh frozen plasma (FFP) is one of the most frequently ordered blood products in the emergency department—and one of the most commonly misused.
Abnormal numbers make us uncomfortable.
Bleeding patients make us anxious.
FFP often feels like the safest middle ground.
Yet evidence over the last two decades consistently shows that FFP corrects laboratory values far more reliably than it improves patient-centred outcomes (Duguid, 2004; Nascimento, 2010).
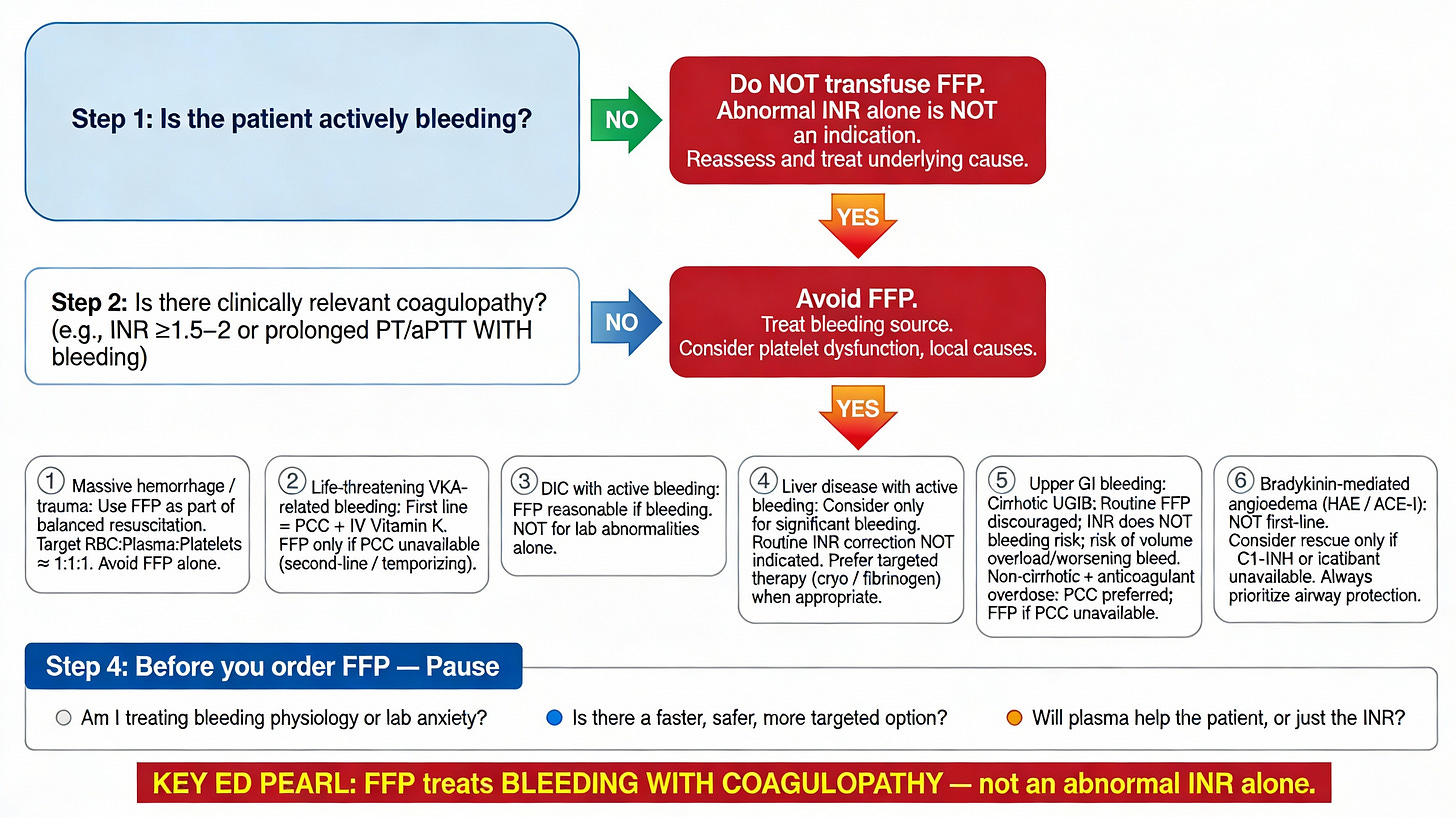
When FFP is indicated in the acute care?
Guidelines, audits, and clinical trials converge on limited, specific indications for FFP in acute care (Duguid, 2004; Nascimento, 2010; Khan, 2018; Biu, 2018).
1. Active Bleeding With Documented Coagulopathy
FFP is appropriate in patients with:
Major trauma or massive hemorrhage
Spontaneous bleeding
Significantly prolonged coagulation parameters (typically INR ≥1.5–2)
This is particularly relevant when prothrombin complex concentrate (PCC) or specific factor concentrates are unavailable (Nascimento, 2010; Prinja, 2017).
Here, FFP functions as part of balanced resuscitation, not as a stand-alone correction of laboratory abnormalities.
2. Disseminated Intravascular Coagulation (DIC) With Bleeding
FFP should be considered only when DIC is accompanied by active bleeding, not for abnormal coagulation tests alone (Duguid, 2004; Khan, 2018).
3. Acute Liver Failure With Active Bleeding or High-Risk Emergency Procedures
In patients with liver failure, FFP may be reasonable when:
There is ongoing bleeding, or
An urgent, high-risk procedure cannot be delayed
Routine INR correction in stable cirrhosis offers no proven benefit (Nascimento, 2010; Biu, 2018).
4. Thrombotic Thrombocytopenic Purpura (TTP)
FFP is essential as part of plasma exchange therapy.
In the ED, its role is stabilization and early initiation while arranging definitive care (Duguid, 2004).
5. Life-Threatening Bleeding - anticoagulants associated (Vit K antagonists)
FFP may be used for:
Intracranial hemorrhage
Massive gastrointestinal bleeding
Only when PCC is unavailable, and always alongside vitamin K (Duguid, 2004; Khan, 2018; Biu, 2018).
The Truth About FFP in Upper GI Bleeding
Upper gastrointestinal bleeding (UGIB) is one of the most common scenarios where FFP is ordered reflexively—but evidence consistently shows this practice is rarely helpful.
In cirrhotic patients with UGIB, FFP transfusion does not reduce active or recent bleeding at endoscopy, even when multiple units are administered (Baxi, 2017). In broader UGIB populations, FFP use has been associated with higher 90-day mortality, particularly when INR is <1.5, without any reduction in rebleeding (Liu, 2022). Randomized trials in high-risk peptic ulcer bleeding further show that adding FFP to endoscopic epinephrine therapy does not improve hemostasis, rebleeding rates, need for surgery, or mortality compared with epinephrine alone (Khodadoostan, 2016).
A central explanation lies in the concept of “rebalanced hemostasis” in cirrhosis. Although INR is prolonged, both pro-coagulant and anti-coagulant factors are reduced, meaning INR does not reflect true bleeding risk (Tripodi & Mannucci, 2011; Northup & Caldwell, 2013). FFP only partially and transiently corrects INR, without restoring effective thrombin generation (Stanworth, 2011; Tripodi, 2017).
Beyond lack of benefit, FFP may actually worsen bleeding in cirrhotic UGIB. Large plasma volumes increase intravascular volume, portal venous pressure, and variceal wall tension, potentially exacerbating hemorrhage rather than controlling it (Bosch, 2008). In addition, FFP is associated with TRALI, TACO, pulmonary edema, and acute lung injury, complications particularly dangerous in cirrhotic patients with limited cardiopulmonary reserve (Gajic, 2006; Dara, 2005).
When coagulation support is truly required, targeted therapy is often more physiologic. Hypofibrinogenemia is common during severe bleeding, and fibrinogen replacement with cryoprecipitate or fibrinogen concentrate addresses a key substrate for clot formation without the excessive volume load of FFP (Tripodi, 2017; Itagaki, 2023). Viscoelastic testing (TEG/ROTEM) consistently shows that many cirrhotic UGIB patients have preserved clot strength despite abnormal INR, allowing clinicians to safely avoid unnecessary plasma transfusion (Kumar, 2020).
An Important Exception: Non-Cirrhotic UGIB With Anticoagulant Overdose
These data should not be misapplied to non-cirrhotic UGIB caused by anticoagulant excess, particularly vitamin K antagonist–related bleeding. In patients with life-threatening UGIB due to warfarin overdose, reversal of coagulopathy is essential. PCC remains the preferred therapy, but FFP is an acceptable second-line option when PCC or specific reversal agents are unavailable, especially in resource-limited settings, and should always be administered alongside vitamin K (Duguid, 2004; Karaca, 2014; Khan, 2018). Even here, FFP should be viewed as a temporizing measure, not default therapy.
Bottom line for UGIB: FFP should not be routine, particularly in cirrhosis. It should be reserved for patients with true coagulopathy and ongoing, significant bleeding, ideally guided by clinical context or viscoelastic testing (Nascimento, 2010).
FFP in Bradykinin-Mediated Angioedema: A Rescue Option
FFP also has a limited but important role in bradykinin-mediated angioedema, including hereditary angioedema (HAE) and ACE-inhibitor–associated angioedema. FFP provides C1-esterase inhibitor and other kinin-degrading enzymes, which can reduce bradykinin levels and lead to clinical improvement in severe attacks, including laryngeal edema (Prematta, 2007; Tang, 2012; Wentzel, 2019).
However, FFP acts slower and less predictably than targeted therapies such as plasma-derived or recombinant C1-INH and icatibant, and carries the usual transfusion-related risks, including volume overload and allergic reactions (Stanworth, 2007; Pacific, 2023). Current expert consensus therefore considers FFP a second-line or rescue therapy, appropriate only when targeted agents are unavailable, and always alongside early airway vigilance and definitive airway planning (Longhurst, 2018; Valerieva & Longhurst, 2022).
When FFP Should Not Be Used
Strong evidence advises against FFP in the following situations:
Mild to moderate INR elevation (INR <1.5–1.8) without bleeding (Duguid, 2004; Khan, 2018)
Prophylactic use before low-risk procedures (central venous catheter, chest tube, tracheostomy)
Randomized trials show no reduction in bleeding (Müller, 2015)
DIC without bleeding (Lauzier, 2007; Biu, 2018)
Volume expansion or nutritional supplementation
Warfarin reversal without severe, active bleeding (Khan, 2018)
Audits from ED and ICU settings consistently show that 40–60% of FFP transfusions are inappropriate, driven mainly by prophylactic or lab-triggered use (Emektar, 2016; Prinja, 2017; Shah, 2020).
Treat physiology.
Respect evidence.
Transfuse with intent.
Caution / Disclaimer
This content is intended for educational and reflective purposes only.
It is designed to support learning, discussion, and clinical reasoning—not to replace formal guidelines, institutional protocols, or bedside clinical judgment.
Patient management decisions must always be individualised and made in real time, based on the clinical context, available resources, and senior/consultant guidance.
References
Duguid J, Atterbury C, Maggs P, et al. Guidelines for the use of fresh‐frozen plasma, cryoprecipitate and cryosupernatant. Br J Haematol. 2004;126(1):11-28.
Nascimento B, Callum J, Rubenfeld G, et al. Clinical review: fresh frozen plasma in massive bleeding—more questions than answers. Crit Care. 2010;14(1):202.
Khan M, Khan M, Dar A, et al. Fresh frozen plasma: indications and misuse. Prof Med J. 2018;25(7):1090-1095.
Biu E, Beraj S, Vyshka G, et al. Transfusion of fresh frozen plasma in critically ill patients: effective or useless? Open Access Maced J Med Sci. 2018;6(4):820-823.
Prinja N, Sharma S, Narain R. Fresh frozen plasma utilization pattern in a tertiary care hospital. Int J Res Med Sci. 2017;5(12):5372-5376.
Emektar E, Dağar S, Çorbacıoğlu Ş, et al. Audit of fresh frozen plasma usage in the emergency department. Turk J Emerg Med. 2016;16(4):137-140.
Shah S, Sidhu M, Gupta V. Audit of fresh frozen plasma usage: a prospective study. Int J Res Med Sci. 2020;8(4):1371-1376.
Lauzier F, Cook D, Griffith L, et al. Fresh frozen plasma transfusion in critically ill patients. Crit Care Med. 2007;35(7):1655-1659.
Müller MC, Arbous MS, Spoelstra-de Man AME, et al. Transfusion of fresh frozen plasma before invasive procedures in critically ill patients: a randomized clinical trial. Transfusion. 2015;55(1):26-35.
Upper Gastrointestinal Bleeding & Cirrhosis
Baxi A, Grant S, Teng B, et al. Impact of fresh frozen plasma transfusion on upper gastrointestinal bleeding in patients with cirrhosis. Am J Gastroenterol. 2017;112(Suppl 1):S305-S306.
Liu S, Zhang X, Walline J, et al. Fresh frozen plasma in acute upper gastrointestinal bleeding does not improve outcomes. Front Med (Lausanne). 2022;9:934024.
Khodadoostan M, Karami-Horestani M, Shavakhi A, et al. Endoscopic treatment for high-risk bleeding peptic ulcers: epinephrine alone vs epinephrine plus fresh frozen plasma. J Res Med Sci. 2016;21:123.
Rebalanced Hemostasis & INR Limitations
Tripodi A, Mannucci PM. The coagulopathy of chronic liver disease. N Engl J Med. 2011;365(2):147-156.
Northup PG, Caldwell SH. Coagulation in liver disease: a guide for the clinician. Clin Gastroenterol Hepatol. 2013;11(9):1064-1074.
Stanworth SJ. The evidence-based use of fresh-frozen plasma. Hematology Am Soc Hematol Educ Program. 2007:179-186.
Stanworth SJ, Walsh TS, Prescott RJ, et al. A national study of plasma use in critical care. Crit Care. 2011;15(2):R108.
Tripodi A. Hemostasis in cirrhosis: rebalanced but fragile. Semin Thromb Hemost. 2017;43(5):455-462.
Portal Pressure, Volume Overload & Transfusion Harm
Bosch J, Abraldes JG, Berzigotti A, et al. Portal hypertension and gastrointestinal bleeding. Semin Liver Dis. 2008;28(1):3-25.
Gajic O, Dzik WH, Toy P. Fresh frozen plasma and platelet transfusion for non-bleeding patients. Crit Care Med. 2006;34(5 Suppl):S170-S173.
Dara SI, Rana R, Afessa B, et al. Fresh frozen plasma transfusion in critically ill medical patients. Crit Care Med. 2005;33(11):2667-2671.
Fibrinogen, Cryoprecipitate & Viscoelastic Testing
Itagaki Y, Hayakawa M, Takahashi Y, et al. Emergency administration of fibrinogen concentrate for hemorrhage. World J Emerg Surg. 2023;18:42.
Kumar M, Ahmad J, Maiwall R, et al. Thromboelastography-guided blood component use in cirrhosis with non-variceal bleeding. Hepatology. 2020;71(1):235-246.
Anticoagulant-Related GI Bleeding
Karaca M, Erbil B, Ozmen M. Prothrombin complex concentrate vs fresh frozen plasma in warfarin-related GI bleeding. Am J Emerg Med. 2014;32(6):660-664.
Bradykinin-Mediated Angioedema
Prematta MJ, Gibbs JG, Pratt EL, et al. Fresh frozen plasma for hereditary angioedema. Ann Allergy Asthma Immunol. 2007;98(4):383-388.
Tang R, Chen S, Zhang H. Fresh frozen plasma for treatment of hereditary angioedema attacks. Chin Med Sci J. 2012;27(2):92-95.
Wentzel N, Panieri A, Ayazi M, et al. Fresh frozen plasma for on-demand hereditary angioedema treatment. World Allergy Organ J. 2019;12(11):100049.
Longhurst H. Optimum use of acute treatments for hereditary angioedema. Front Med (Lausanne). 2018;4:245.
Valerieva A, Longhurst H. Treatment of hereditary angioedema—pathways to rescue. Front Allergy. 2022;3:952233.
Pacific A, Jindal A, Sil A, et al. Management of hereditary angioedema in resource-constrained settings. Asia Pac Allergy. 2023;13(1):60-65.



Nice summary covering all patients seen in the ED where FFP is used. I have a comment regarding a subgroup that has not been addressed. These general statements do not hold good for snake bite victims. FFP early after ASV administration in hemotoxic snake bite even without signs of bleeding have shown faster resolution of coagulopathy. Read this systematic review - https://bmjopen.bmj.com/content/15/12/e102745