Beyond INR: How TEG and ROTEM Changed Bleeding Resuscitation
From blind transfusion to physiology‑guided hemostasis; Bleeding Isn’t Just About INR
For decades, when a bleeding patient rolled in, we reached for the same tools:
• INR • PT/aPTT • Platelet count
And then we transfused — often blindly.
Plasma for a high INR. Platelets for a low count. Cryo when things looked bad.
But anyone who’s managed trauma, cirrhosis, ECMO, or massive hemorrhage knows the truth:
👉 Conventional labs don’t tell the full coagulation story.
Enter TEG (Thromboelastography) and ROTEM (Rotational Thromboelastometry) — viscoelastic, whole‑blood, point‑of‑care tests that show clot formation in real time.
Not just numbers.
Actual physiology.
Why Traditional Coagulation Tests Fall Short
Standard labs measure isolated parts of the coagulation cascade.
They don’t show:
❌ Platelet function ❌ Clot strength ❌ Fibrinolysis ❌ The balance between clotting and anticoagulation
And they’re slow.
In active bleeding, minutes matter.
TEG and ROTEM give actionable results in 5–15 minutes.
TEG vs ROTEM: Similar Physiology, Different Strengths
While TEG and ROTEM are built on the same viscoelastic principles, there are practical differences that influence bedside use. ROTEM generally provides faster early clot formation parameters (CT, CA5, CA10) and more robust assessment of fibrinogen contribution through FIBTEM, making it particularly useful for early detection of hypofibrinogenemia in trauma hemorrhage (Baksaas-Aasen et al., 2019; Maegele, 2024).
TEG, on the other hand, is highly sensitive to clot initiation abnormalities and has been widely studied in goal-directed massive transfusion protocols (Gonzalez et al., 2016; Gonzalez et al., 2017).
Comparative studies suggest ROTEM may return actionable data a few minutes earlier, while both platforms perform similarly in guiding transfusion decisions and outcomes (Brill et al., 2021; Ines et al., 2025).
Importantly, no high-quality evidence demonstrates clear clinical superiority of one system over the other—both enable physiology-guided resuscitation that outperforms conventional coagulation testing in complex bleeding states (Whiting & Dinardo, 2014; Bugaev et al., 2020).
What Makes TEG So Powerful (Strengths)
TEG provides an integrated view of hemostasis:
✅ Assesses interactions between clotting factors, platelets, and endogenous anticoagulants
✅ Measures platelet function, not just platelet count
✅ Evaluates clot strength and fibrinogen contribution
✅ Detects hyperfibrinolysis (missed by standard labs)
✅ Rapidly identifies heparin effect
✅ Reduces INR‑driven plasma overuse (one of its biggest clinical wins)
Evidence shows TEG‑guided transfusion can reduce unnecessary platelet and plasma use while maintaining hemostasis.
In short:
👉 Smarter blood. Less guesswork.
Where TEG Falls Short (Weaknesses)
It’s not magic.
TEG is limited in detecting:
⚠️ Most antiplatelet drugs (aspirin, clopidogrel — unless platelet mapping is used)
⚠️ DOACs (apixaban, rivaroxaban, dabigatran)
⚠️ von Willebrand disease
⚠️ Hypothermia and hypocalcemia effects (since testing occurs at 37°C)
⚠️ Rare coagulation disorders
It also can’t precisely identify specific factor deficiencies.
👉 Think of TEG as a global physiology tool, not a diagnostic microscope.
Where TEG & ROTEM Shine in Acute Care
They’re most useful when multiple coagulation abnormalities occur simultaneously:
🔥 Trauma‑induced coagulopathy
🔥 DIC
🔥 Cirrhosis‑related bleeding
🔥 ECMO
In these complex states, traditional labs often mislead.
TEG/ROTEM show what the clot is actually doing.
A Practical Way to Read TEG/ROTEM at the Bedside (Without Getting Lost)
Viscoelastic assays generate many numbers — but in acute bleeding, only a few parameters drive meaningful decisions.
Think in four physiologic questions:
👉 Is clotting starting on time?
👉 Is the clot strong enough?
👉 Is fibrinogen adequate?
👉 Is the clot breaking down too fast?
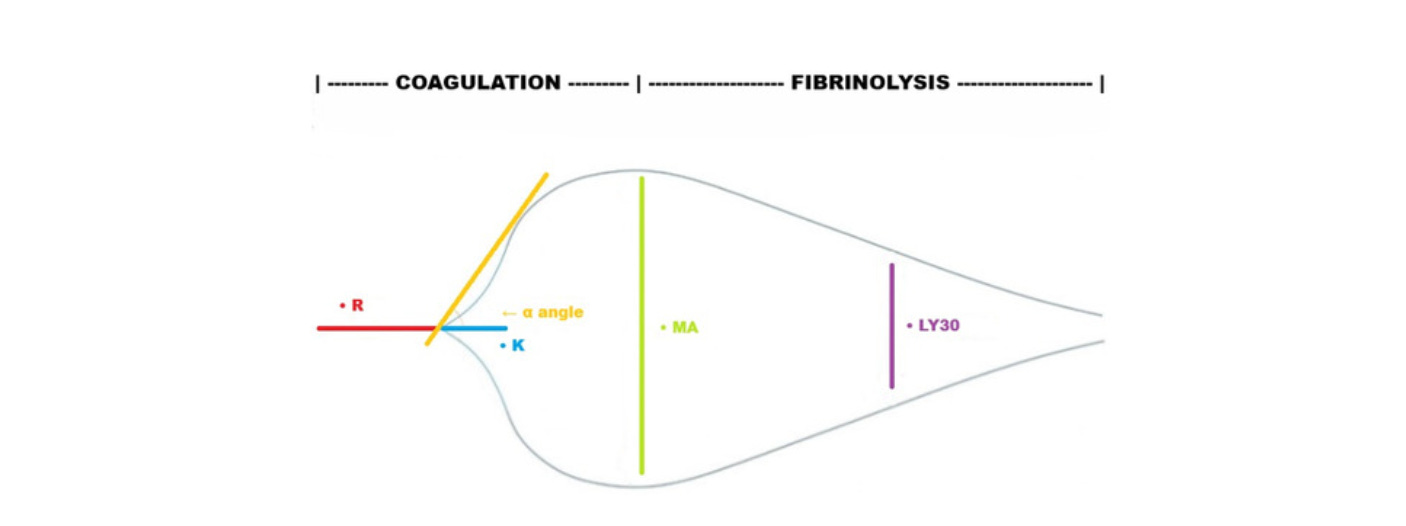
1️⃣ Clot Initiation → R-Time (TEG) / CT (ROTEM)
⏳ Prolonged = clotting factor deficiency or heparin effect
✔️ Normal = enzymatic coagulation is intact (plasma usually not needed even if INR is high)
Clinical use: • Give plasma or PCC if markedly prolonged and bleeding
• Check heparinase channel if available to rule out heparin effect
2️⃣ Clot Formation Speed → Alpha Angle (TEG) / CFT & CA5–CA10 (ROTEM)
📐 Reflects how quickly the clot strengthens (driven mainly by fibrinogen with platelet contribution)
Low values suggest: • Hypofibrinogenemia • Poor clot propagation
Clinical pearl: 👉 This is an early warning sign — falling angle/CA5 often precedes low clot strength.
3️⃣ Clot Strength → MA (TEG) / MCF (ROTEM)
This is the most actionable parameter.
Low MA/MCF indicates weak clot due to:
🧩 Low fibrinogen
🧩 Platelet dysfunction or low count
How to differentiate: • Use functional fibrinogen (TEG 6S / FIBTEM on ROTEM) • If fibrinogen low → give fibrinogen/cryo first
• If fibrinogen normal but clot weak → give platelets
4️⃣ Fibrinogen Contribution → Functional Fibrinogen (TEG) / FIBTEM (ROTEM)
Isolates the fibrin-based clot component.
Low values = true fibrinogen deficiency (common in trauma and massive hemorrhage).
👉 Often the earliest and most important target in bleeding resuscitation.
5️⃣ Clot Breakdown → LY30 (TEG) / Lysis Index (ROTEM)
📈 Elevated = hyperfibrinolysis → consider TXA or antifibrinolytics
⚠️ Normal does not always exclude fibrinolysis (especially with older TEG systems)
Quick Bedside Algorithm
• Prolonged R/CT → plasma or PCC
• Low angle or CA5 → think fibrinogen early
• Low MA/MCF → fibrinogen ± platelets
• High lysis → TXA
Heparin Effect Check
If R‑time improves with heparinase → heparin (exogenous or auto‑heparinization) is contributing.
Important in:
• Sepsis • Trauma • ECMO
What About TEG 6S and ROTEM?
Newer systems (like TEG 6S) improve:
✔️ Fibrinogen measurement ✔️ Heparin neutralization ✔️ Faster workflows
ROTEM is similarly powerful and often quicker in fibrinogen detection.
Both integrate seamlessly into massive transfusion protocols.
What Does the Evidence Actually Show?
Across trauma and major bleeding studies:
📚 TEG/ROTEM detect coagulopathy earlier than standard labs
📚 Better predict massive transfusion needs
📚 Identify fibrinolysis missed by routine testing
📚 Guide targeted blood product use
Systematic reviews suggest:
✅ Reduced plasma and platelet transfusion
⚠️ Possible survival benefit (but evidence quality is low‑moderate)
Most strong RCT data comes from cardiac surgery — ED‑specific randomized trials are still limited.
But trauma observational data consistently supports improved hemostatic precision.
TEG/ROTEM in Cirrhotic UGI & Variceal Bleeding: Why It Matters
In cirrhosis, INR and platelet counts poorly reflect true hemostatic balance, and empiric correction can worsen portal pressures and volume overload. Viscoelastic testing has emerged as a physiology‑guided alternative for acute UGI bleeding. In a randomized trial of cirrhotics with non‑variceal UGI bleeding, TEG‑guided transfusion significantly reduced use of FFP, platelets, and cryoprecipitate without increasing rebleeding or mortality (Kumar et al., 2020).
Similar reductions in blood product exposure have been observed with ROTEM‑based strategies in critically ill cirrhotics (Gopal et al., 2024) and across meta‑analyses of viscoelastic‑guided correction in cirrhosis (Shenoy et al., 2022).
In acute variceal hemorrhage, TEG guidance not only minimized transfusion requirements but was associated with lower 42‑day rebleeding compared with conventional lab‑guided care (Rout et al., 2020), and contemporary reviews now support viscoelastic‑based approaches in severe coagulopathy (Edelson et al., 2021). Together, these data suggest TEG/ROTEM allow safer, more restrictive transfusion in cirrhotic bleeding without compromising hemostasis.
TEG/ROTEM in Trauma Hemorrhage: From Fixed Ratios to Physiology-Guided Resuscitation
In major trauma, up to one-third of patients develop trauma-induced coagulopathy (TIC), which is strongly associated with increased mortality (Gonzalez et al., 2017; Brill et al., 2021; Maegele, 2024).
Viscoelastic testing rapidly characterizes clot initiation, strength, fibrinogen contribution, and fibrinolysis within minutes, allowing early identification of hypofibrinogenemia and hyperfibrinolysis that are often missed or delayed by PT/INR-based testing (Da Luz et al., 2014; Hunt et al., 2015; Baksaas-Aasen et al., 2019). Randomized trials have shown that TEG-guided massive transfusion strategies can improve early survival and reduce unnecessary plasma and platelet exposure compared with conventional laboratory-guided resuscitation (Gonzalez et al., 2016; Gonzalez et al., 2017), though larger multicenter studies such as ITACTIC found no clear mortality benefit overall, highlighting ongoing uncertainty (Baksaas-Aasen et al., 2020).
Meta-analyses and trauma guidelines nevertheless support viscoelastic-guided algorithms as adjuncts to standard massive transfusion protocols, consistently demonstrating reduced blood product use with possible outcome benefit and no signal of harm (Bugaev et al., 2020; Wikkelsø et al., 2017).
The Real Takeaway for Emergency & Critical Care
TEG and ROTEM don’t replace clinical judgment.
But they transform how we resuscitate bleeding patients.
Instead of asking:
❓ “What’s the INR?”
We ask:
✅ Is clotting delayed? ✅ Is the clot weak? ✅ Is fibrinolysis happening?
And we treat exactly what’s wrong.
Bottom Line
TEG and ROTEM offer:
✔️ Rapid, real‑time coagulation physiology
✔️ Targeted transfusion strategies
✔️ Less blind product use
✔️ Better understanding of complex coagulopathies
They’re most valuable in:
🩸 Trauma 🩸 Massive hemorrhage 🩸 Cirrhosis 🩸 ECMO 🩸 DIC
And less useful for simple anticoagulation monitoring.
If you work in acute care and still rely solely on INR to guide transfusion — it’s time to rethink.
Share this with your trauma team, residents, or ICU colleagues.
More evidence‑based ED physiology coming soon at Life on the Frontline.
References
Whiting D, Dinardo J. TEG and ROTEM: technology and clinical applications. Am J Hematol. 2014;89(2):228-232. doi:10.1002/ajh.23599
Da Luz LT, Nascimento B, Shankarakutty AK, Rizoli S, Adhikari NKJ. Effect of thromboelastography and rotational thromboelastometry on diagnosis of coagulopathy, transfusion guidance, and mortality in trauma: a systematic review. Crit Care. 2014;18(5):518. doi:10.1186/s13054-014-0518-9
Gonzalez E, Moore EE, Moore HB. Management of trauma-induced coagulopathy with thromboelastography. Crit Care Clin. 2017;33(1):119-134. doi:10.1016/j.ccc.2016.09.002
Brill JB, Brenner M, Duchesne J, et al. The role of TEG and ROTEM in damage control resuscitation. Shock. 2021;56(1S):52-61. doi:10.1097/SHK.0000000000001686
Wikkelsø A, Wetterslev J, Møller AM, Afshari A. Thromboelastography or thromboelastometry to monitor haemostatic treatment versus usual care in bleeding patients. Cochrane Database Syst Rev. 2016;(8):CD007871. doi:10.1002/14651858.CD007871.pub3
Bugaev N, Como JJ, Golani G, et al. Thromboelastography and rotational thromboelastometry in bleeding patients with coagulopathy: EAST practice management guideline. J Trauma Acute Care Surg. 2020;89(6):999-1017. doi:10.1097/TA.0000000000002944
Schmidt AE, Israel AK, Refaai MA. The utility of thromboelastography to guide blood product transfusion. Am J Clin Pathol. 2019;152(4):407-422. doi:10.1093/ajcp/aqz074
Kvisselgaard AD, Wolthers S, Wikkelsø A, et al. TEG/ROTEM-guided algorithms in bleeding patients: updated systematic review and meta-analysis. Acta Anaesthesiol Scand. 2024;69(1):e14558. doi:10.1111/aas.14558
Drumheller BC, Stein DM, Moore LJ, et al. Thromboelastography and rotational thromboelastometry for the surgical intensivist. J Trauma Acute Care Surg. 2019;86(4):710-721. doi:10.1097/TA.0000000000002206
Rech MA, Gilbert B, Nei SD, et al. How to use viscoelastic testing in critical illness. J Am Coll Clin Pharm. 2023;6(8):954-963. doi:10.1002/jac5.1829
Kumar M, Ahmad J, Maiwall R, et al. Thromboelastography-guided blood component use in patients with cirrhosis with nonvariceal bleeding: a randomized controlled trial. Hepatology. 2020;71(1):235-246. doi:10.1002/hep.30794
Gopal N, Panwar S, Saluja V, et al. Impact of coagulopathy assessment with thromboelastography and thromboelastometry on transfusion requirements in critically ill cirrhosis with nonvariceal bleeding. Int J Crit Illn Inj Sci. 2024;14(2):67-73. doi:10.4103/ijciis.ijciis_65_23
Shenoy A, Louissaint J, Shannon C, Tapper EB, Lok ASF. Viscoelastic testing prior to nonsurgical procedures reduces blood product use without increasing bleeding risk in cirrhosis. Dig Dis Sci. 2022;67(12):5290-5299. doi:10.1007/s10620-021-07376-6
Rout G, Gunjan D, Mahapatra SJ, et al. Thromboelastography-guided blood product transfusion in cirrhosis patients with variceal bleeding. J Clin Gastroenterol. 2020;54(3):255-262. doi:10.1097/MCG.0000000000001214
Edelson J, Basso J, Rockey DC. Updated strategies in the management of acute variceal haemorrhage. Curr Opin Gastroenterol. 2021;37(3):167-172. doi:10.1097/MOG.0000000000000723
Hunt H, Stanworth S, Curry N, et al. Thromboelastography and rotational thromboelastometry for trauma-induced coagulopathy. Cochrane Database Syst Rev. 2015;(2):CD010438. doi:10.1002/14651858.CD010438.pub2
Baksaas-Aasen K, van Dieren S, Balvers K, et al. Data-driven development of ROTEM and TEG algorithms for trauma hemorrhage management. Ann Surg. 2019;270(6):1178-1185. doi:10.1097/SLA.0000000000002825
Gonzalez E, Moore EE, Moore HB, et al. Goal-directed hemostatic resuscitation of trauma-induced coagulopathy: a randomized clinical trial. Ann Surg. 2016;263(6):1051-1059. doi:10.1097/SLA.0000000000001608
Baksaas-Aasen K, Gall L, Stensballe J, et al. Viscoelastic haemostatic assay–augmented protocols for major trauma haemorrhage (ITACTIC): a randomized controlled trial. Intensive Care Med. 2021;47(1):49-59. doi:10.1007/s00134-020-06266-1
Wikkelsø A, Wetterslev J, Møller AM, Afshari A. Thromboelastography or thromboelastometry to monitor haemostatic treatment: systematic review with meta-analysis. Anaesthesia. 2017;72(4):519-531. doi:10.1111/anae.13765
Maegele M. Update on the pathophysiology and management of trauma-induced coagulopathy based on viscoelastic testing. Clin Exp Emerg Med. 2024;11(3):259-267. doi:10.15441/ceem.24.202