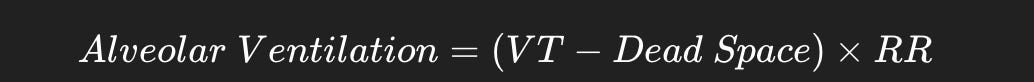Acute Hypercapnia: A Mechanistic Approach to Ventilator Troubleshooting
Distinguishing central hypoventilation from mechanical airflow limitation to guide safe, physiology-based ventilator adjustments.
Hypercapnia is not a diagnosis.
It is a physiologic signal:
Alveolar ventilation is inadequate relative to CO₂ production.
Formally:
(VT = Tidal volume)
When PaCO₂ rises, one (or more) of the following is happening:
Minute ventilation is insufficient
Dead space is increased
Expiratory flow is limited
CO₂ production has increased beyond ventilatory capacity
Before adjusting the ventilator, the most important question is:
Is this a drive problem — or a mechanics problem?
Because the ventilator strategy differs completely.
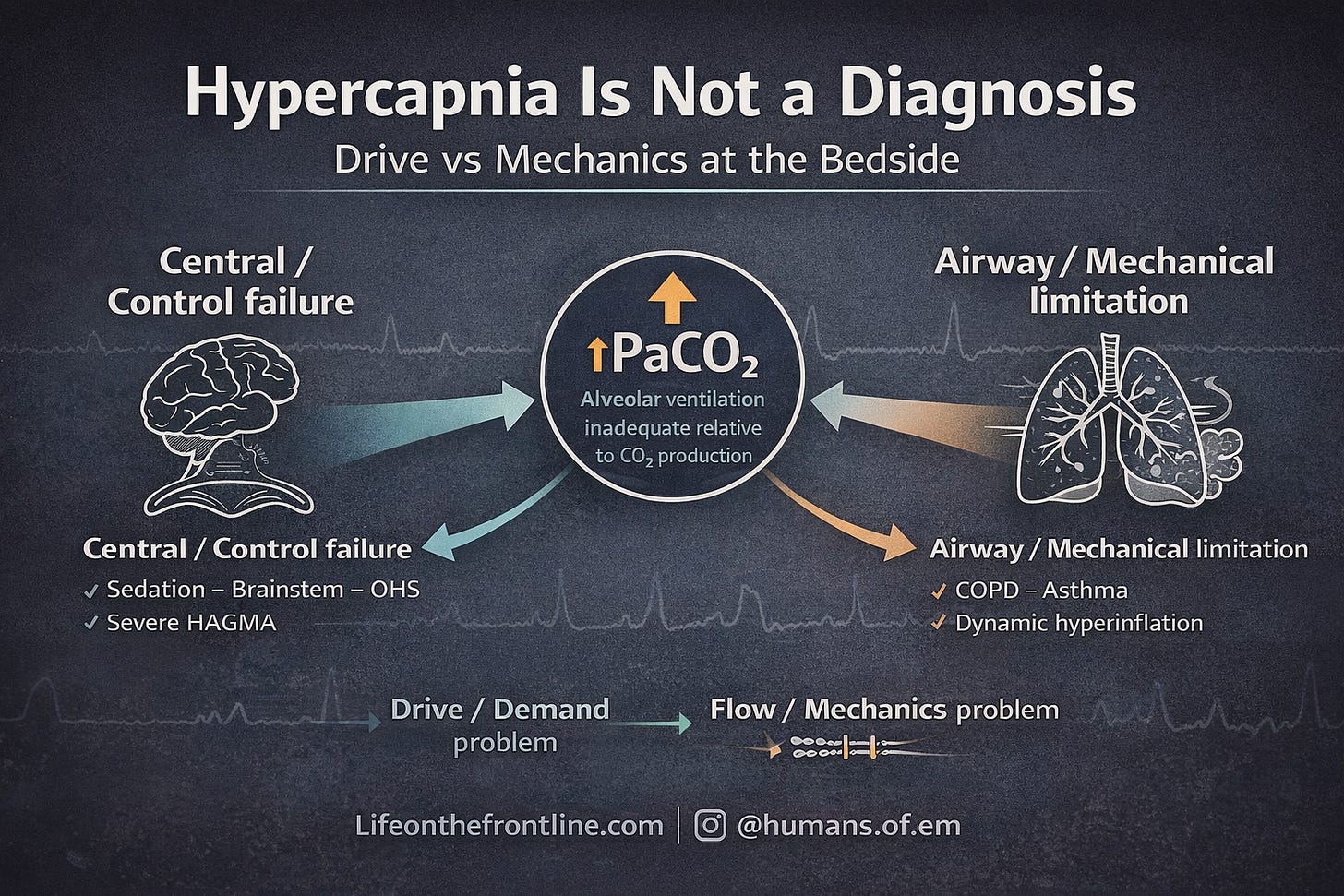
Central vs Airway Causes of Hypercapnia
The Classification That Changes Everything
Hypercapnia reflects inadequate alveolar ventilation relative to CO₂ production (Roussos & Koutsoukou, 2003; Csoma et al., 2022).
Broadly, causes fall into two groups:
1. Central (Control) — “Won’t Breathe”
2. Airway / Mechanical (Pump) — “Can’t Breathe”
This distinction determines ventilator management.
TYPE 1 — CENTRAL HYPERVENTILATORY FAILURE
1.Absent or Insufficient Respiratory Drive
In central causes:
Lungs structurally normal
Compliance preserved
Airways patent
The problem is:
Absent respiratory drive — or insufficient respiratory drive for the metabolic demand of the setting.
Hypercapnia develops because minute ventilation is reduced, not because lungs are damaged (Nanayakkara & McNamara, 2024).
A. Depressed CNS Drive
Common causes:
Opioids
Sedatives
Benzodiazepines
General anesthesia
Drug overdose
Population data confirm these as leading causes of hypercapnic respiratory failure (Chung et al., 2023).
Mechanistic discussions are provided by Brown (2010).
B. Structural Brainstem Injury
Brainstem stroke
CNS infection
Head trauma
Post-anoxic injury
These impair the medullary respiratory centers (Roussos & Koutsoukou, 2003).
C. Primary Central Hypoventilation Syndromes
Congenital Central Hypoventilation Syndrome
ROHHAD
Genetic disorders with blunted CO₂ chemosensitivity
Reviewed by Kasi & Perez (2024) and Amin (2021).
These patients may compensate while awake but fail during sleep or sedation.
D. Obesity Hypoventilation Syndrome (Central Component)
OHS is not purely mechanical.
There is:
Blunted hypercapnic ventilatory response
Reduced chemosensitivity
Leptin resistance
Discussed extensively by Masa et al. (2019) and Amorim et al. (2022).
E. Insufficient Ventilation for Disease Demand
The patient may have respiratory drive — but not enough to meet metabolic demand.
Example:
Severe high-anion-gap metabolic acidosis (HAGMA)
If compensatory hyperventilation is inadequate → hypercapnia supervenes.
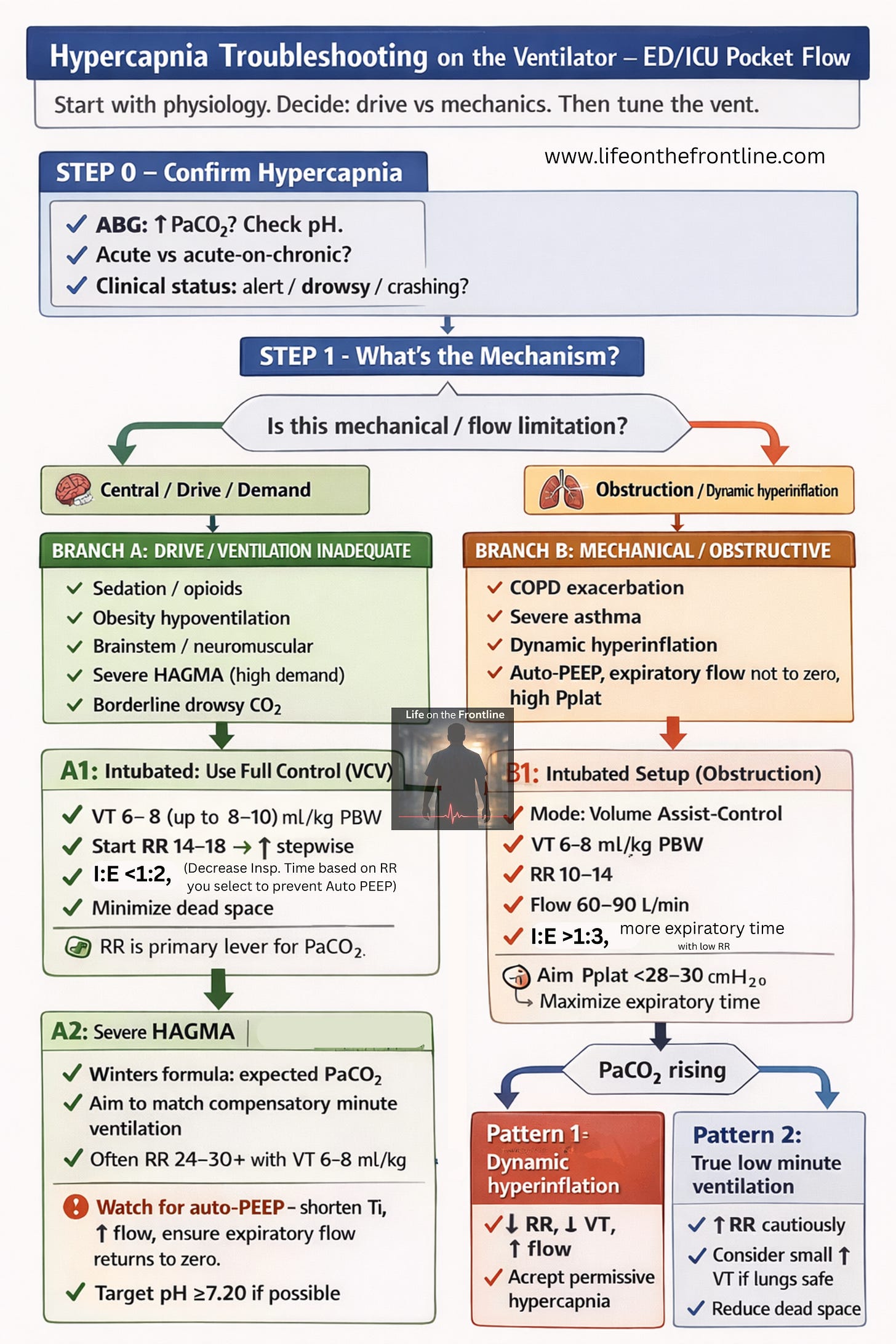
Ventilator Strategy in Central Causes
Use Full Control Mode (VCV)
Do not rely on:
Pressure support alone
Spontaneous modes
You must guarantee alveolar ventilation.
Tiruvoipati et al. (2020) emphasize that controlled ventilation is necessary when drive is unreliable.
Core Settings (Structurally Normal Lungs)
Tidal Volume (VT)
6–8 mL/kg predicted body weight
May increase toward 8–10 mL/kg if plateau pressure acceptable
(Almanza-Hurtado et al., 2022)
Respiratory Rate (RR)
Start 14–18/min
Increase stepwise to normalize PaCO₂
RR is the primary lever
I:E Ratio
~1:2 unless obstruction present
Dead Space
Remove unnecessary connectors
Avoid excessive apparatus dead space
(Zuiki et al., 2020)
Monitoring
Because drive is absent:
Serial ABGs
Continuous end-tidal CO₂
Transcutaneous CO₂ where appropriate
(Khayat et al., 2017)
Permissive hypercapnia is not the strategy here.
You are replacing a failed respiratory control system.
SPECIAL SCENARIO — SEVERE HAGMA WITH PaCO₂ RETENTION
In severe metabolic acidosis:
The primary threat is very low pH, not CO₂ itself.
Patients compensate via extreme hyperventilation (Kussmaul breathing).
If intubation reduces this compensation:
PaCO₂ rises
pH drops further
Hemodynamic collapse may follow
Severe hypercapnic acidosis is associated with worse outcomes (Nin et al., 2017; Tiruvoipati et al., 2017).
Step 1 — Calculate Target PaCO₂
Use Winter’s formula:
This defines expected compensation.
Not 40 mmHg.
Step 2 — Set Initial Minute Ventilation
Based on ABG-guided strategy:
Severe HAGMA often requires MV > 15–20 L/min.
Step 3 — Initial Ventilator Settings
Mode: VCV
VT: 6–8 mL/kg PBW
RR: 24–30/min (or higher)
Increase RR preferentially
ABG within 15–30 minutes
Guided by principles outlined by Tiruvoipati et al. (2020) and Achanti & Szerlip (2022).
Step 4 — Adjust Using Goal MV Formula
Increase MV accordingly.
MV = RR * VT
Consider RR as primary lever, avoid increasing VT >8ml/kg (IBW)
Critical Practical Insight
When RR > 24/min:
Expiratory time shortens
Auto-PEEP may develop even in normal lungs
If auto-PEEP appears:
Decrease inspiratory time
Increase inspiratory flow
Ensure expiratory flow returns to baseline before next breath
Failure to do this can convert a central problem into a mechanical one.
Targets
Aim for pH ≥ 7.20
Avoid sudden PaCO₂ rise
Frequent ABGs
In severe metabolic acidosis: Permissive hypercapnia can be dangerous.
TYPE 2 — OBSTRUCTIVE / MECHANICAL HYPERCAPNIA
“Can’t Breathe”
In COPD/asthma:
Hypercapnia results from:
Airflow limitation
Dynamic hyperinflation
Intrinsic PEEP
Increased dead space
(Csoma et al., 2022; Shigemura et al., 2020)
The danger is air trapping — not the CO₂ number itself.
Initial Strategy
Mode: Volume Assist–Control
VT: 6–8 mL/kg PBW
RR: 10–14/min
Flow: 60–90 L/min
I:E: 1:3–1:4
Pplat < 28–30 cmH₂O
(Davidson et al., 2016; Demoule et al., 2020)
Goal:
Maximize expiratory time.
Accept moderate hypercapnia if pH acceptable.
Pattern-Based Troubleshooting
Pattern 1
↑ PaCO₂ + High Pplat + Auto-PEEP
→ Dynamic hyperinflation
Management:
↓ RR
↓ VT
↑ Inspiratory flow
Accept permissive hypercapnia
(Demoule et al., 2020)
Pattern 2
↑ PaCO₂ + Normal Pressures
→ True low minute ventilation
Management:
Increase RR cautiously
Increase VT if safe
Reduce dead space
(Tiruvoipati et al., 2020)
Pattern 3
Severe Hypercapnia Despite Protective Settings
Consider:
Alternative ventilator strategies.
What Is AVAPS / iVAPS in Non-Invasive Ventilation?
AVAPS = Average Volume-Assured Pressure Support
iVAPS = Intelligent Volume-Assured Pressure Support
Both are hybrid NIV modes that combine:
Pressure support ventilation
With a target tidal volume
In standard BiPAP (S/T mode):
You set IPAP (inspiratory pressure)
You set EPAP
The delivered tidal volume depends on:
Patient effort
Lung mechanics
Leaks
The machine delivers fixed pressure.
In AVAPS/VAPS:
You set a target tidal volume.
The ventilator automatically adjusts inspiratory pressure to achieve it.
So instead of “fixed pressure → variable VT”, you get:
Variable pressure → relatively stable VT
That is the key difference.
How AVAPS / iVAPS Works Physiologically
You typically set:
Target VT (e.g., 6–8 mL/kg PBW)
EPAP
Minimum IPAP
Maximum IPAP
Backup rate
The ventilator:
Measures delivered tidal volume
Compares it to the target
Gradually increases or decreases IPAP
Keeps VT close to the preset goal
It does this over several breaths (not breath-to-breath like invasive volume control, but dynamically over time).
So AVAPS behaves like:
A semi-volume-controlled NIV mode.
Why This Matters in Hypercapnia
Hypercapnia improves when alveolar ventilation improves.
In hypercapnic patients on NIV, TV is the most unstable variable.
If TV falls → CO₂ rises.
AVAPS stabilizes TV.
That stabilizes alveolar ventilation.
That improves CO₂ clearance.
Why It’s Particularly Helpful in Borderline Drowsy Patients
Consider the “gray-zone” patient:
Acute COPD exacerbation
PaCO₂ 75 mmHg
pH 7.23
Drowsy but arousable
This patient:
Has fluctuating respiratory drive
Has inconsistent effort
May drift into hypoventilation
With standard BiPAP:
If effort drops → VT drops → CO₂ rises → mental status worsens → more hypoventilation.
You must manually increase IPAP.
With AVAPS:
If effort drops → VT drops → machine increases IPAP (within limits) → VT preserved → CO₂ continues to wash out.
This provides:
A safety buffer
Smoother CO₂ correction
Less frequent manual retitration
Claudett et al. (2013) showed faster PaCO₂ and GCS improvement in hypercapnic encephalopathy using AVAPS compared to standard S/T mode.
Importantly:
Hard outcomes (intubation, mortality) are generally similar to well-managed conventional BiPAP (Gören et al., 2021; Evans et al., 2024).
So AVAPS improves physiologic stability — not necessarily survival.
When AVAPS Is Most Useful
AVAPS is particularly helpful in:
Borderline drowsy hypercapnic patients
Fluctuating respiratory drive
Fatigue suspected
NIV started early in COPD exacerbation
“Almost intubation” scenarios - can help in preventilation during preparation for intubation
It buys time while:
Bronchodilators work
Steroids reduce inflammation
CO₂ levels gradually fall
When It Is Less Useful
Massive mask leak
Severe agitation
Immediate intubation criteria
Profound hemodynamic instability
Severe HAGMA requiring very high minute ventilation
AVAPS is still NIV — it cannot replace invasive control when compensation demands are extreme.
Practical AVAPS Setup in Acute Hypercapnia
Typical initial approach:
Target VT: 6–8 mL/kg PBW
EPAP: 4–6 cmH₂O (adjust for oxygenation)
Min IPAP: 10–12 cmH₂O
Max IPAP: 20–25 cmH₂O
Backup RR: 12–16/min
Reassess:
Clinical status
ABG at 1–2 hours
Mental status
Work of breathing
Bedside Algorithm to tackle - courtesy - Life on the Frontline , humans.of.em - Insta
⚠️ For educational purposes only. In case of doubt, follow your institutional protocols and consult your seniors.
References
Roussos C, Koutsoukou A. Respiratory failure. Eur Respir J. 2003;22(suppl 47):3s-14s. doi:10.1183/09031936.03.00038503
Nanayakkara B, McNamara S. Pathophysiology of chronic hypercapnic respiratory failure. Sleep Med Clin. 2024;19(3):379-389. doi:10.1016/j.jsmc.2024.04.001
Brown LK. Hypoventilation syndromes. Clin Chest Med. 2010;31(2):249-270. doi:10.1016/j.ccm.2010.03.002
Chung Y, Garden F, Marks G, Vedam H. Causes of hypercapnic respiratory failure: a population-based case-control study. BMC Pulm Med. 2023;23:Article 26. doi:10.1186/s12890-023-02639-6
Kasi A, Perez I. Congenital central hypoventilation syndrome and disorders of control of ventilation. Clin Chest Med. 2024;45(3):663-673. doi:10.1016/j.ccm.2024.02.018
Amin R. Beyond the retrotrapezoid nucleus in congenital central hypoventilation syndrome. Am J Respir Crit Care Med. 2021;205(3):271-272. doi:10.1164/rccm.202111-2602ed
Masa JF, Pépin JL, Borel JC, Mokhlesi B, Murphy PB, Sánchez-Quiroga MÁ. Obesity hypoventilation syndrome. Eur Respir Rev. 2019;28(151):180097. doi:10.1183/16000617.0097-2018
Amorim M, Aung O, Mokhlesi B, Polotsky VY. Leptin-mediated neural targets in obesity hypoventilation syndrome. Sleep. 2022;45(10):zsac153. doi:10.1093/sleep/zsac153
Tiruvoipati R, Gupta S, Pilcher D, Bailey M. Management of hypercapnia in critically ill mechanically ventilated patients—A narrative review of literature. J Intensive Care Soc. 2020;21(4):327-333. doi:10.1177/1751143720915666
Tiruvoipati R, Pilcher D, Buscher H, Botha J, Bailey M. Effects of hypercapnia and hypercapnic acidosis on hospital mortality in mechanically ventilated patients. Crit Care Med. 2017;45(7):e649-e656. doi:10.1097/CCM.0000000000002332
Nin N, Muriel A, Peñuelas Ó, et al. Severe hypercapnia and outcome of mechanically ventilated patients with moderate or severe ARDS. Intensive Care Med. 2017;43(2):200-208. doi:10.1007/s00134-016-4611-1
Achanti A, Szerlip HM. Acid-base disorders in the critically ill patient. Clin J Am Soc Nephrol. 2022;17(4):595-607. doi:10.2215/CJN.04500422
Grasselli G, Calfee CS, Camporota L, et al. ESICM guidelines on acute respiratory distress syndrome: definition, phenotyping and respiratory support strategies. Intensive Care Med. 2023;49(7):727-759. doi:10.1007/s00134-023-07050-7
Zuiki M, Naito Y, Kitamura K, et al. Reduction in minute alveolar ventilation causes hypercapnia in ventilated neonates with respiratory distress. Eur J Pediatr. 2021;180(1):241-246. doi:10.1007/s00431-020-03761-x
Csoma B, Vulpi M, Dragonieri S, et al. Hypercapnia in COPD: causes, consequences, and therapy. J Clin Med. 2022;11(11):3180. doi:10.3390/jcm11113180
Shigemura M, Homma T, Sznajder JI. Hypercapnia: an aggravating factor in asthma. J Clin Med. 2020;9(10):3207. doi:10.3390/jcm9103207
Davidson AC, Banham S, Elliott M, et al. BTS/ICS guideline for the ventilatory management of acute hypercapnic respiratory failure in adults. Thorax. 2016;71(suppl 2):ii1-ii35. doi:10.1136/thoraxjnl-2015-208209
Demoule A, Brochard L, Dres M, et al. How to ventilate obstructive and asthmatic patients. Intensive Care Med. 2020;46(12):2436-2449. doi:10.1007/s00134-020-06291-0
Osadnik CR, Tee VS, Carson-Chahhoud KV, et al. Non-invasive ventilation for the management of acute hypercapnic respiratory failure due to exacerbation of COPD. Cochrane Database Syst Rev. 2017;(7):CD004104. doi:10.1002/14651858.CD004104.pub4
Claudett KH, Claudett M, Wong M, et al. Noninvasive mechanical ventilation with average volume assured pressure support in patients with COPD and hypercapnic encephalopathy. BMC Pulm Med. 2013;13:12. doi:10.1186/1471-2466-13-12
Gören N, Şancı E, Coşkun F, et al. Comparison of BPAP S/T and average volume-assured pressure support modes for hypercapnic respiratory failure in the emergency department: a randomized controlled trial. Balkan Med J. 2021;38(5):265-271. doi:10.5152/balkanmedj.2021.20137
Evans A, Alam A, Nugent K. Average volume-assured pressure support versus fixed pressure support in chronic hypercapnic respiratory failure: a systematic review and meta-analysis. Southwest Respir Crit Care Chron. 2024;12(53). doi:10.12746/swrccc.v12i53.1383
Macrea M, Oczkowski S, Rochwerg B, et al. Long-term noninvasive ventilation in chronic stable hypercapnic COPD: an official ATS clinical practice guideline. Am J Respir Crit Care Med. 2020;202(4):e74-e87. doi:10.1164/rccm.202006-2382ST
Giraud R, Banfi C, Assouline B, et al. The use of extracorporeal CO₂ removal in acute respiratory failure. Ann Intensive Care. 2021;11:43. doi:10.1186/s13613-021-00824-6